Targeted Nanotechnology in Glioblastoma Multiforme
- PMID: 28408882
- PMCID: PMC5374154
- DOI: 10.3389/fphar.2017.00166
Targeted Nanotechnology in Glioblastoma Multiforme
Abstract
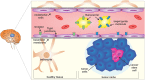
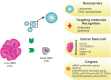
Gliomas, and in particular glioblastoma multiforme, are aggressive brain tumors characterized by a poor prognosis and high rates of recurrence. Current treatment strategies are based on open surgery, chemotherapy (temozolomide) and radiotherapy. However, none of these treatments, alone or in combination, are considered effective in managing this devastating disease, resulting in a median survival time of less than 15 months. The efficiency of chemotherapy is mainly compromised by the blood-brain barrier (BBB) that selectively inhibits drugs from infiltrating into the tumor mass. Cancer stem cells (CSCs), with their unique biology and their resistance to both radio- and chemotherapy, compound tumor aggressiveness and increase the chances of treatment failure. Therefore, more effective targeted therapeutic regimens are urgently required. In this article, some well-recognized biological features and biomarkers of this specific subgroup of tumor cells are profiled and new strategies and technologies in nanomedicine that explicitly target CSCs, after circumventing the BBB, are detailed. Major achievements in the development of nanotherapies, such as organic poly(propylene glycol) and poly(ethylene glycol) or inorganic (iron and gold) nanoparticles that can be conjugated to metal ions, liposomes, dendrimers and polymeric micelles, form the main scope of this summary. Moreover, novel biological strategies focused on manipulating gene expression (small interfering RNA and clustered regularly interspaced short palindromic repeats [CRISPR]/CRISPR associated protein 9 [Cas 9] technologies) for cancer therapy are also analyzed. The aim of this review is to analyze the gap between CSC biology and the development of targeted therapies. A better understanding of CSC properties could result in the development of precise nanotherapies to fulfill unmet clinical needs.
Keywords: blood–brain barrier; cancer stem cell; glioma; nanomedicine; nanotechnology; targeted therapy.
Figures

References
-
- Albrecht K. W., De Witt Hamer P. C., Leenstra S., Bakker P. J., Beijnen J. H., Troost D., et al. (2001). High concentration of Daunorubicin and Daunorubicinol in human malignant astrocytomas after systemic administration of liposomal Daunorubicin. J. Neurooncol. 53 267–271. 10.1023/A:1012287212388 - DOI - PubMed
-
- American Cancer Society (2016). Cancer Statistics Center. Available at: https://cancerstatisticscenter.cancer.org/?_ga=1.14923958.1099926323.148... (accessed October 2016).
-
- Ananda S., Nowak A. K., Cher L., Dowling A., Brown C., Simes J., et al. (2011). Phase 2 trial of temozolomide and pegylated liposomal doxorubicin in the treatment of patients with glioblastoma multiforme following concurrent radiotherapy and chemotherapy. J. Clin. Neurosci. 18 1444–1448. 10.1016/j.jocn.2011.02.026 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

