Glycogen phosphorylase inhibition improves beta cell function
- PMID: 28409826
- PMCID: PMC5758390
- DOI: 10.1111/bph.13819
Glycogen phosphorylase inhibition improves beta cell function
Abstract
Background and purpose: Glycogen phosphorylase (GP) is the key enzyme for glycogen degradation. GP inhibitors (GPi-s) are glucose lowering agents that cause the accumulation of glucose in the liver as glycogen. Glycogen metabolism has implications in beta cell function. Glycogen degradation can maintain cellular glucose levels, which feeds into catabolism to maintain insulin secretion, and elevated glycogen degradation levels contribute to glucotoxicity. The purpose of this study was to assess whether influencing glycogen metabolism in beta cells by GPi-s affects the function of these cells.
Experimental approach: The effects of structurally different GPi-s were investigated on MIN6 insulinoma cells and in a mouse model of diabetes.
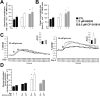
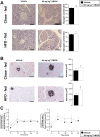
Key results: GPi treatment increased glycogen content and, consequently, the surface area of glycogen in MIN6 cells. Furthermore, GPi treatment induced insulin receptor β (InsRβ), Akt and p70S6K phosphorylation, as well as pancreatic and duodenal homeobox 1(PDX1) and insulin expression. In line with these findings, GPi-s enhanced non-stimulated and glucose-stimulated insulin secretion in MIN6 cells. The InsRβ was shown to co-localize with glycogen particles as confirmed by in silico screening, where components of InsR signalling were identified as glycogen-bound proteins. GPi-s also activated the pathway of insulin secretion, indicated by enhanced glycolysis, mitochondrial oxidation and calcium signalling. Finally, GPi-s increased the size of islets of Langerhans and improved glucose-induced insulin release in mice.
Conclusion and implications: These data suggest that GPi-s also target beta cells and can be repurposed as agents to preserve beta cell function or even ameliorate beta cell dysfunction in different forms of diabetes.
Linked articles: This article is part of a themed section on Inventing New Therapies Without Reinventing the Wheel: The Power of Drug Repurposing. To view the other articles in this section visit http://onlinelibrary.wiley.com/doi/10.1111/bph.v175.2/issuetoc.
© 2017 The British Pharmacological Society.
Figures

References
-
- Abdul‐Ghani MA (2013). Type 2 diabetes and the evolving paradigm in glucose regulation. The American journal of managed care 19: S43–50. - PubMed
-
- Agius L (2007). New hepatic targets for glycaemic control in diabetes. Best Pract Res Clin Endocrinol Metab 21: 587–605. - PubMed
-
- Agius L (2015). Role of glycogen phosphorylase in liver glycogen metabolism. Mol Aspects Med 46: 34–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

