Position and Orientation Insensitive Wireless Power Transmission for EnerCage-Homecage System
- PMID: 28410095
- PMCID: PMC5619985
- DOI: 10.1109/TBME.2017.2691720
Position and Orientation Insensitive Wireless Power Transmission for EnerCage-Homecage System
Abstract
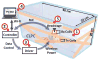
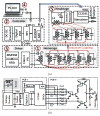
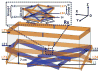
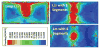
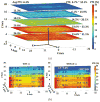
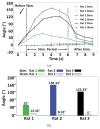
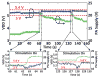
We have developed a new headstage architecture as part of a smart experimental arena, known as the EnerCage-HC2 system, which automatically delivers stimulation and collects behavioral data over extended periods with minimal small animal subject handling or personnel intervention in a standard rodent homecage. Equipped with a four-coil inductive link, the EnerCage-HC2 system wirelessly powers the receiver (Rx) headstage, irrespective of the subject's location or head orientation, eliminating the need for tethering or carrying bulky batteries. On the transmitter (Tx) side, a driver coil, five high-quality (Q) factor segmented resonators at different heights and orientations, and a closed-loop Tx power controller create a homogeneous electromagnetic (EM) field within the homecage 3-D space, and compensate for drops in power transfer efficiency (PTE) due to Rx misalignments. The headstage is equipped with four small slanted resonators, each covering a range of head orientations with respect to the Tx resonators, which direct the EM field toward the load coil at the bottom of the headstage. Moreover, data links based on Wi-Fi, UART, and Bluetooth low energy are utilized to enables remote communication and control of the Rx. The PTE varies within 23.6%-33.3% and 6.7%-10.1% at headstage heights of 8 and 20 cm, respectively, while continuously delivering >40 mW to the Rx electronics even at 90° rotation. As a proof of EnerCage-HC2 functionality in vivo, a previously documented on-demand electrical stimulation of the globus pallidus, eliciting consistent head rotation, is demonstrated in three freely behaving rats.
Figures











References
-
- Grill WM, Norman SE, Bellamkonda RV. Implanted neural interfaces: biochallenges and engineered solutions. Annual review of biomedical engineering. 2009 Aug;11:1–24. - PubMed
-
- Donoghue JP. Bridging the brain to the world: a perspective on neural interface systems. Neuron. 2008 Nov;60:511–521. - PubMed
-
- Aravanis AM, et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. Journal of neural engineering. 2007 Jun;4:S143. - PubMed
-
- Shaw FZ, Lai CJ, Chiu TH. A low-noise flexible integrated system for recording and analysis of multiple electrical signals during sleep–wake states in rats. Journal of neuroscience methods. 2002 Jul;118:77–87. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

