Elements including metals in the atomizer and aerosol of disposable electronic cigarettes and electronic hookahs
- PMID: 28414730
- PMCID: PMC5393578
- DOI: 10.1371/journal.pone.0175430
Elements including metals in the atomizer and aerosol of disposable electronic cigarettes and electronic hookahs
Abstract
Objective: Our purpose was to quantify 36 inorganic chemical elements in aerosols from disposable electronic cigarettes (ECs) and electronic hookahs (EHs), examine the effect of puffing topography on elements in aerosols, and identify the source of the elements.
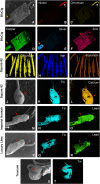
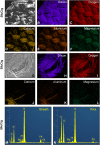
Methods: Thirty-six inorganic chemical elements and their concentrations in EC/EH aerosols were determined using inductively coupled plasma optical emission spectroscopy, and their source was identified by analyzing disassembled atomizers using scanning electron microscopy and energy dispersive X-ray spectroscopy.
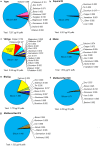
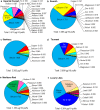
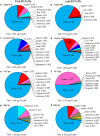
Results: Of 36 elements screened, 35 were detected in EC/EH aerosols, while only 15 were detected in conventional tobacco smoke. Some elements/metals were present in significantly higher concentrations in EC/EH aerosol than in cigarette smoke. Concentrations of particular elements/metals within EC/EH brands were sometimes variable. Aerosols generated at low and high air-flow rates produced the same pattern of elements, although the total element concentration decreased at the higher air flow rate. The relative amount of elements in the first and last 60 puffs was generally different. Silicon was the dominant element in aerosols from all EC/EH brands and in cigarette smoke. The elements appeared to come from the filament (nickel, chromium), thick wire (copper coated with silver), brass clamp (copper, zinc), solder joints (tin, lead), and wick and sheath (silicon, oxygen, calcium, magnesium, aluminum). Lead was identified in the solder and aerosol of two brands of EHs (up to 0.165 μg/10 puffs).
Conclusion: These data show that EC/EH aerosols contain a mixture of elements, including heavy metals, with concentrations often significantly higher than in conventional cigarette smoke. While the health effects of inhaling mixtures of heated metals is currently not known, these data will be valuable in future risk assessments involving EC/EH elements/metals.
Conflict of interest statement
Figures



References
-
- Williams M, Villarreal A, Bozhilov K, Lin S, Talbot P. Metal and silicate particles including nanoparticles are present in electronic cigarette cartomizer fluid and aerosol. PLoS One. 2013;8(3): e57987 doi: 10.1371/journal.pone.0057987 - DOI - PMC - PubMed
-
- Williams M, To A, Bozhilov K, Talbot P. Strategies to Reduce Tin and Other Metals in Electronic Cigarette Aerosol. PLoS One. 2015;10(9): e0138933 doi: 10.1371/journal.pone.0138933 - DOI - PMC - PubMed
-
- Goniewicz ML, Knysak J, Gawron M, Kosmider L, Sobczak A, Kurek J, et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob Control. 2014;23(2): 133–9. doi: 10.1136/tobaccocontrol-2012-050859 - DOI - PMC - PubMed
-
- Laugesen M. Safety Report on the Ruyan ® e-cigarette Cartridge and Inhaled Aerosol. 2008.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

