Inhibition of iron overload-induced apoptosis and necrosis of bone marrow mesenchymal stem cells by melatonin
- PMID: 28415572
- PMCID: PMC5458235
- DOI: 10.18632/oncotarget.16382
Inhibition of iron overload-induced apoptosis and necrosis of bone marrow mesenchymal stem cells by melatonin
Abstract
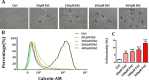
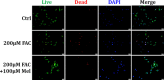
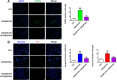
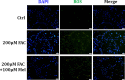
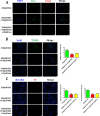
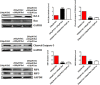
Iron overload induces severe damage to several vital organs such as the liver, heart and bone, and thus contributes to the dysfunction of these organs. The aim of this study is to investigate whether iron overload causes the apoptosis and necrosis of bone marrow mesenchymal stem cells (BMSCs) and melatonin may prevent its toxicity. Perls' Prussion blue staining showed that exposure to increased concentrations of ferric ammonium citrate (FAC) induced a gradual increase of intracellular iron level in BMSCs. Trypan blue staining demonstrated that FAC decreased the viability of BMSCs in a concentration-dependent manner. Notably, melatonin protected BMSCs against apoptosis and necrosis induced by FAC and it was vertified by Live/Dead, TUNEL and PI/Hoechst stainings. Furthermore, melatonin pretreatment suppressed FAC-induced reactive oxygen species accumulation. Western blot showed that exposure to FAC resulted in the decrease of anti-apoptotic protein Bcl-2 and the increase of pro-apoptotic protein Bax and Cleaved Caspase-3, and necrosis-related proteins RIP1 and RIP3, which were significantly inhibited by melatonin treatment. At last, melatonin receptor blocker luzindole failed to block the protection of BMSCs apoptosis and necrosis by melatonin. Taken together, melatonin protected BMSCs from iron overload induced apoptosis and necrosis by regulating Bcl-2, Bax, Cleaved Caspase-3, RIP1 and RIP3 pathways.
Keywords: apoptosis; bone marrow mesenchymal stem cells; iron overload; melatonin; necrosis.
Conflict of interest statement
The authors had no conflicts of interest to declare in relation to this article.
Figures

References
-
- Sekiya I, Larson BL, Vuoristo JT, Cui JG, Prockop DJ. Adipogenic differentiation of human adult stem cells from bone marrow stroma (MSCs) J Bone Miner Res. 2004;19:256–264. - PubMed
-
- Yang YJ, Qian HY, Huang J, Geng YJ, Gao RL, Dou KF, Yang GS, Li JJ, Shen R, He ZX, Lu MJ, Zhao SH. Atorvastatin treatment improves survival and effects of implanted mesenchymal stem cells in post-infarct swine hearts. Eur Heart J. 2008;29:1578–1590. - PubMed
-
- Wang A, Shen F, Liang Y, Wang J. Marrow-derived MSCs and atorvastatin improve cardiac function in rat model of AMI. Int J Cardiol. 2011;150:28–32. - PubMed
-
- Dong Q, Yang Y, Song L, Qian H, Xu Z. Atorvastatin prevents mesenchymal stem cells from hypoxia and serum-free injury through activating AMP-activated protein kinase. Int J Cardiol. 2011;153:311–316. - PubMed
-
- Noronha-Matos JB, Coimbra J, Sa-e-Sousa A, Rocha R, Marinhas J, Freitas R, Guerra-Gomes S, Ferreirinha F, Costa MA, Correia-de-Sa P. P2×7-induced zeiosis promotes osteogenic differentiation and mineralization of postmenopausal bone marrow-derived mesenchymal stem cells. FASEB J. 2014;28:5208–5222. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

