Selenophosphate synthetase 1 (SPS1) is required for the development and selenium homeostasis of central nervous system in chicken (Gallus gallus)
- PMID: 28415800
- PMCID: PMC5482627
- DOI: 10.18632/oncotarget.16283
Selenophosphate synthetase 1 (SPS1) is required for the development and selenium homeostasis of central nervous system in chicken (Gallus gallus)
Abstract
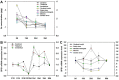
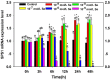
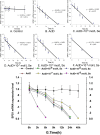
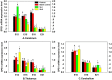
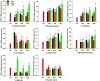
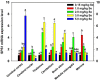
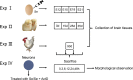
Selenophosphate synthetase (SPS) is essential for selenoprotein biosynthesis. In two SPS paralogues, SPS1 was only cloned from a cDNA library prepared from avian organ. However, the biological function of SPS1 in chicken central nervous system (CNS) remains largely unclear. To investigate the role of avian SPS1 in the development and selenium (Se) homeostasis of CNS, fertile eggs, chicken embryos, embryo neurons and chicks were employed in this study. The response of SPS1 transcription to the development and Se levels of CNS tissues was analyzed using qRT-PCR. SPS1 gene exists extensively in the development of chicken CNS. The wide expression of avian SPS1 can be controlled by the Se content levels, which suggests that SPS1 is important in the regulation of Se homeostasis. The fundamental mechanism of these effects is that Se alters the half-life and stability of SPS1 mRNA. Therefore, SPS1 exerts an irreplaceable biological function in chicken CNS and Se homeostasis is closely related to the expression of SPS1. These results suggested that SPS1 was required for the development and Se homeostasis of CNS in chicken.
Keywords: central nervous system; chicken; development; selenium homeostasis; selenophosphate synthetase 1.
Conflict of interest statement
The authors declare that there are no conflicts of interests.
Figures

Similar articles
-
Prediction of selenoprotein T structure and its response to selenium deficiency in chicken immune organs.Biol Trace Elem Res. 2014 Aug;160(2):222-31. doi: 10.1007/s12011-014-0049-x. Epub 2014 Jun 25. Biol Trace Elem Res. 2014. PMID: 24958020
-
Effects of dietary selenium deficiency or excess on gene expression of selenoprotein N in chicken muscle tissues.Biol Trace Elem Res. 2014 Mar;157(3):234-41. doi: 10.1007/s12011-014-9893-y. Epub 2014 Feb 2. Biol Trace Elem Res. 2014. PMID: 24488809 Clinical Trial.
-
Selenophosphate synthetase 1 is an essential protein with roles in regulation of redox homoeostasis in mammals.Biochem J. 2016 Jul 15;473(14):2141-54. doi: 10.1042/BCJ20160393. Epub 2016 May 16. Biochem J. 2016. PMID: 27208177 Free PMC article.
-
The selenophosphate synthetase family: A review.Free Radic Biol Med. 2022 Nov 1;192:63-76. doi: 10.1016/j.freeradbiomed.2022.09.007. Epub 2022 Sep 16. Free Radic Biol Med. 2022. PMID: 36122644 Review.
-
Utilization of selenocysteine as a source of selenium for selenophosphate biosynthesis.Biofactors. 2001;14(1-4):69-74. doi: 10.1002/biof.5520140110. Biofactors. 2001. PMID: 11568442 Review.
Cited by
-
Selenomethionine Attenuated H2O2-Induced Oxidative Stress and Apoptosis by Nrf2 in Chicken Liver Cells.Antioxidants (Basel). 2023 Aug 29;12(9):1685. doi: 10.3390/antiox12091685. Antioxidants (Basel). 2023. PMID: 37759988 Free PMC article.
References
-
- Brauer AU, Savaskan NE. Molecular actions of selenium in the brain: neuroprotective mechanisms of an essential trace element. Rev Neurosci. 2004;15:19–32. - PubMed
-
- Savaskan NE, Bräuer AU, Kühbacher M, Eyüpoglu IY, Kyriakopoulos A, Ninnemann O, Behne D, Nitsch R. Selenium deficiency increases susceptibility to glutamate-induced excitotoxicity. FASEB J. 2003;17:112–4. - PubMed
-
- Vinceti M, Maraldi T, Bergomi M, Malagoli C. Risk of chronic low-dose selenium overexposure in humans: insights from epidemiology and biochemistry. Rev Environ Health. 2009;24:231–48. - PubMed
-
- Maraldi T, Riccio M, Zambonin L, Vinceti M, De Pol A, Hakim G. Low levels of selenium compounds are selectively toxic for a human neuron cell line through ROS/RNS increase and apoptotic process activation. Neurotoxicology. 2011;32:180–7. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

