Regulatory functional territory of PLK-1 and their substrates beyond mitosis
- PMID: 28415805
- PMCID: PMC5514964
- DOI: 10.18632/oncotarget.16290
Regulatory functional territory of PLK-1 and their substrates beyond mitosis
Abstract
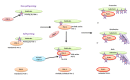
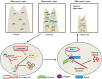
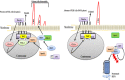
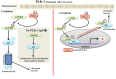
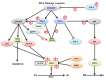
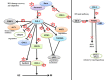
Polo-like kinase 1 (PLK-1) is a well-known (Ser/Thr) mitotic protein kinase and is considered as a proto-oncogene. As hyper-activation of PLK-1 is broadly associated with poor prognosis and cancer progression, it is one of the most extensively studied mitotic kinases. During mitosis, PLK-1 regulates various cell cycle events, such as spindle pole maturation, chromosome segregation and cytokinesis. However, studies have demonstrated that the role of PLK-1 is not only restricted to mitosis, but PLK-1 can also regulate other vital events beyond mitosis, including transcription, translation, ciliogenesis, checkpoint adaptation and recovery, apoptosis, chromosomes dynamics etc. Recent reviews have tried to define the regulatory role of PLK-1 during mitosis progression and tumorigenesis, but its' functional role beyond mitosis is still largely unexplored. PLK-1 can regulate the activity of many proteins that work outside of its conventional territory. The dysregulation of these proteins can cause diseases such as Alzheimer's disease, tumorigenesis etc. and may also lead to drug resistance. Thus, in this review, we discussed the versatile role of PLK-1 and tried to collect data to validate its' functional role in cell cycle regulation apart from mitosis.
Keywords: DNA damage response; checkpoint adaptation and recovery; ciliogenesis; polo-like kinase-1; transcription and translation.
Conflict of interest statement
There is no author conflict to disclose.
Figures
Similar articles
-
PLK-1: Angel or devil for cell cycle progression.Biochim Biophys Acta. 2016 Apr;1865(2):190-203. doi: 10.1016/j.bbcan.2016.02.003. Epub 2016 Feb 18. Biochim Biophys Acta. 2016. PMID: 26899266 Review.
-
The polo-like kinase PLKA is required for initiation and progression through mitosis in the filamentous fungus Aspergillus nidulans.Mol Microbiol. 2005 Jan;55(2):572-87. doi: 10.1111/j.1365-2958.2004.04404.x. Mol Microbiol. 2005. PMID: 15659171
-
Polo-like kinase in trypanosomes: an odd member out of the Polo family.Open Biol. 2020 Oct;10(10):200189. doi: 10.1098/rsob.200189. Epub 2020 Oct 14. Open Biol. 2020. PMID: 33050792 Free PMC article.
-
Regulation of Polo-like kinase 1 by DNA damage in mitosis. Inhibition of mitotic PLK-1 by protein phosphatase 2A.J Biol Chem. 2007 Jan 26;282(4):2473-82. doi: 10.1074/jbc.M605480200. Epub 2006 Nov 22. J Biol Chem. 2007. PMID: 17121863
-
Polo-like kinases and DNA damage checkpoint: beyond the traditional mitotic functions.Exp Biol Med (Maywood). 2011 Jun 1;236(6):648-57. doi: 10.1258/ebm.2011.011011. Epub 2011 May 9. Exp Biol Med (Maywood). 2011. PMID: 21558091 Review.
Cited by
-
SKA3 Promotes Cell Growth in Breast Cancer by Inhibiting PLK-1 Protein Degradation.Technol Cancer Res Treat. 2020 Jan-Dec;19:1533033820947488. doi: 10.1177/1533033820947488. Technol Cancer Res Treat. 2020. PMID: 32799774 Free PMC article.
-
PLK1 as a cooperating partner for BCL2-mediated antiapoptotic program in leukemia.Blood Cancer J. 2023 Sep 7;13(1):139. doi: 10.1038/s41408-023-00914-7. Blood Cancer J. 2023. PMID: 37679323 Free PMC article.
-
Modelling the Functions of Polo-Like Kinases in Mice and Their Applications as Cancer Targets with a Special Focus on Ovarian Cancer.Cells. 2021 May 12;10(5):1176. doi: 10.3390/cells10051176. Cells. 2021. PMID: 34065956 Free PMC article. Review.
-
Effects of Sepantronium Bromide (YM-155) on the Whole Transcriptome of MDA-MB-231 Cells: Highlight on Impaired ATR/ATM Fanconi Anemia DNA Damage Response.Cancer Genomics Proteomics. 2018 Jul-Aug;15(4):249-264. doi: 10.21873/cgp.20083. Cancer Genomics Proteomics. 2018. PMID: 29976630 Free PMC article.
-
Unraveling mitotic protein networks by 3D multiplexed epitope drug screening.Mol Syst Biol. 2018 Aug 13;14(8):e8238. doi: 10.15252/msb.20188238. Mol Syst Biol. 2018. PMID: 30104419 Free PMC article.
References
-
- Llamazares S, Moreira A, Tavares A, Girdham C, Spruce BA, Gonzalez C, Karess RE, Glover DM, Sunkel CE. polo encodes a protein kinase homolog required for mitosis in Drosophila. Genes Dev. 1991;5:2153–2165. - PubMed
-
- Strebhardt K. Multifaceted polo-like kinases: drug targets and antitargets for cancer therapy. Nat Rev Drug Discov. 2010;9:643–660. - PubMed
-
- Sunkel CE, Glover DM. polo, a mitotic mutant of Drosophila displaying abnormal spindle poles. Journal of cell science. 1988;89:25–38. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

