Synthesis and biological evaluation of largazole zinc-binding group analogs
- PMID: 28416100
- PMCID: PMC5494147
- DOI: 10.1016/j.bmc.2017.03.071
Synthesis and biological evaluation of largazole zinc-binding group analogs
Abstract
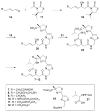
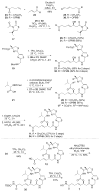
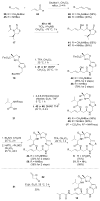
Histone acetylation is an extensively investigated post-translational modification that plays an important role as an epigenetic regulator. It is controlled by histone acetyl transferases (HATs) and histone deacetylases (HDACs). The overexpression of HDACs and consequent hypoacetylation of histones have been observed in a variety of different diseases, leading to a recent focus of HDACs as attractive drug targets. The natural product largazole is one of the most potent natural HDAC inhibitors discovered so far and a number of largazole analogs have been prepared to define structural requirements for its HDAC inhibitory activity. However, previous structure-activity relationship studies have heavily investigated the macrocycle region of largazole, while there have been only limited efforts to probe the effect of various zinc-binding groups (ZBGs) on HDAC inhibition. Herein, we prepared a series of largazole analogs with various ZBGs and evaluated their HDAC inhibition and cytotoxicity. While none of the analogs tested were as potent or selective as largazole, the Zn2+-binding affinity of each ZBG correlated with HDAC inhibition and cytotoxicity. We expect that our findings will aid in building a deeper understanding of the role of ZBGs in HDAC inhibition as well as provide an important basis for the future development of new largazole analogs with non-thiol ZBGs as novel therapeutics for cancer.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures
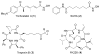
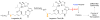

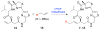

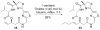
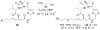
Similar articles
-
Synthesis and HDAC inhibitory activity of isosteric thiazoline-oxazole largazole analogs.Bioorg Med Chem Lett. 2013 Nov 1;23(21):6025-8. doi: 10.1016/j.bmcl.2013.06.012. Epub 2013 Jun 18. Bioorg Med Chem Lett. 2013. PMID: 24035339 Free PMC article.
-
Evaluation of class I HDAC isoform selectivity of largazole analogues.Bioorg Med Chem Lett. 2014 Aug 15;24(16):3728-31. doi: 10.1016/j.bmcl.2014.07.006. Epub 2014 Jul 9. Bioorg Med Chem Lett. 2014. PMID: 25070421 Free PMC article.
-
Largazole Analogues as Histone Deacetylase Inhibitors and Anticancer Agents: An Overview of Structure-Activity Relationships.ChemMedChem. 2017 Dec 7;12(23):1917-1926. doi: 10.1002/cmdc.201700563. Epub 2017 Nov 17. ChemMedChem. 2017. PMID: 29117473 Review.
-
Total synthesis of largazole and analogues: HDAC inhibition, antiproliferative activity and metabolic stability.Bioorg Med Chem. 2011 Jun 15;19(12):3650-8. doi: 10.1016/j.bmc.2011.02.024. Epub 2011 Feb 17. Bioorg Med Chem. 2011. PMID: 21420302
-
Synthetic routes and biological evaluation of largazole and its analogues as potent histone deacetylase inhibitors.Molecules. 2011 Jun 7;16(6):4681-94. doi: 10.3390/molecules16064681. Molecules. 2011. PMID: 21654576 Free PMC article. Review.
Cited by
-
Diversity of small molecule HIV-1 latency reversing agents identified in low- and high-throughput small molecule screens.Med Res Rev. 2020 May;40(3):881-908. doi: 10.1002/med.21638. Epub 2019 Oct 13. Med Res Rev. 2020. PMID: 31608481 Free PMC article. Review.
-
Role of Histone Deacetylases in Carcinogenesis: Potential Role in Cholangiocarcinoma.Cells. 2020 Mar 23;9(3):780. doi: 10.3390/cells9030780. Cells. 2020. PMID: 32210140 Free PMC article. Review.
-
Synthesis and Preliminary Biological Evaluation of Two Fluoroolefin Analogs of Largazole Inspired by the Structural Similarity of the Side Chain Unit in Psammaplin A.Mar Drugs. 2019 Jun 3;17(6):333. doi: 10.3390/md17060333. Mar Drugs. 2019. PMID: 31163697 Free PMC article.
-
Unexpected Enhancement of HDACs Inhibition by MeS Substitution at C-2 Position of Fluoro Largazole.Mar Drugs. 2020 Jun 30;18(7):344. doi: 10.3390/md18070344. Mar Drugs. 2020. PMID: 32629787 Free PMC article.
-
Marine Power on Cancer: Drugs, Lead Compounds, and Mechanisms.Mar Drugs. 2021 Aug 27;19(9):488. doi: 10.3390/md19090488. Mar Drugs. 2021. PMID: 34564150 Free PMC article. Review.
References
-
- Handel AE, Ebers GC, Ramagopalan SV. Epigenetics: molecular mechanisms and implications for disease. Trends Mol Med. 2010;16:7–16. - PubMed
- Arrowsmith CH, Bountra C, Fish PV, Lee K, Schapira M. Epigenetic protein families: a new frontier for drug discovery. Nat Rev Drug Discov. 2012;11:384–400. - PubMed
-
- Falkenberg KJ, Johnstone RW. Histone deacetylases and their inhibitors in cancer, neurological diseases and immune disorders. Nat Rev Drug Discov. 2014;13:673–691. - PubMed
-
- Taori K, Paul VJ, Luesch H. Structure and activity of largazole, a potent antiproliferative agent from the Floridian marine cyanobacterium Symploca sp. J Am Chem Soc. 2008;130:1806–1807. - PubMed
- Engene N, Tronholm A, Salvador-Reyes LA, Luesch H, Paul VJ. Caldora penicillata gen. nov., comb. nov. (cyanobacteria), a pantropical marine species with biomedical relevance. J Phycol. 2015;51:670–681. - PMC - PubMed
- Salvador-Reyes LA, Engene N, Paul VJ, Luesch H. Targeted natural products discovery from marine cyanobacteria using combined phylogenetic and mass spectrometric evaluation. J Nat Prod. 2015;78:486–492. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

