Cisplatin Analogs Confer Protection against Cyanide Poisoning
- PMID: 28416275
- PMCID: PMC5472353
- DOI: 10.1016/j.chembiol.2017.03.013
Cisplatin Analogs Confer Protection against Cyanide Poisoning
Abstract
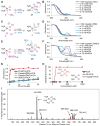
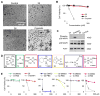
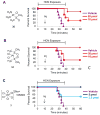
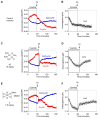
Cisplatin holds an illustrious position in the history of chemistry most notably for its role in the virtual cure of testicular cancer. Here we describe a role for this small molecule in cyanide detoxification in vivo. Cyanide kills organisms as diverse as insects, fish, and humans within seconds to hours. Current antidotes exhibit limited efficacy and are not amenable to mass distribution requiring the development of new classes of antidotes. The binding affinity of the cyanide anion for the positively charged metal platinum is known to create an extremely stable complex in vitro. We therefore screened a panel of diverse cisplatin analogs and identified compounds that conferred protection from cyanide poisoning in zebrafish, mice, and rabbits. Cumulatively, this discovery pipeline begins to establish the characteristics of platinum ligands that influence their solubility, toxicity, and efficacy, and provides proof of concept that platinum-based complexes are effective antidotes for cyanide poisoning.
Keywords: antidote; cisplatin; cyanide; phenotypic screening; platinum; zebrafish.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Conflict of interest statement
There are no conflicts of interest.
Figures


Similar articles
-
Chemical and Clinical Aspects of Metal-Containing Antidotes for Poisoning by Cyanide.Met Ions Life Sci. 2019 Jan 14;19:/books/9783110527872/9783110527872-020/9783110527872-020.xml. doi: 10.1515/9783110527872-020. Met Ions Life Sci. 2019. PMID: 30855115 Review.
-
Disulfides as cyanide antidotes: evidence for a new in vivo oxidative pathway for cyanide detoxification.Chem Res Toxicol. 2009 Dec;22(12):1948-53. doi: 10.1021/tx900258m. Chem Res Toxicol. 2009. PMID: 19891443
-
Nitrocobinamide, a new cyanide antidote that can be administered by intramuscular injection.J Med Chem. 2015 Feb 26;58(4):1750-9. doi: 10.1021/jm501565k. Epub 2015 Feb 16. J Med Chem. 2015. PMID: 25650735 Free PMC article.
-
Which cyanide antidote?Crit Rev Toxicol. 2009;39(7):541-52. doi: 10.1080/10408440802304944. Crit Rev Toxicol. 2009. PMID: 19650716 Review.
-
Antidotes for acute cyanide poisoning.Curr Pharm Biotechnol. 2012 Aug;13(10):1940-8. doi: 10.2174/138920112802273182. Curr Pharm Biotechnol. 2012. PMID: 22352728 Review.
Cited by
-
Redirecting Intermediary Metabolism to Counteract Cyanide Poisoning.FASEB J. 2025 Jun 30;39(12):e70709. doi: 10.1096/fj.202400230RR. FASEB J. 2025. PMID: 40497726 Free PMC article. Review.
-
Investigating the Replacement of Carboxylates with Carboxamides to Modulate the Safety and Efficacy of Platinum(II) Thioether Cyanide Scavengers.Toxicol Sci. 2023 Nov 11;197(2):197-210. doi: 10.1093/toxsci/kfad119. Online ahead of print. Toxicol Sci. 2023. PMID: 37952247 Free PMC article.
-
Identification of specific metabolic pathways as druggable targets regulating the sensitivity to cyanide poisoning.PLoS One. 2018 Jun 7;13(6):e0193889. doi: 10.1371/journal.pone.0193889. eCollection 2018. PLoS One. 2018. PMID: 29879736 Free PMC article.
-
Intramuscular administration of hexachloroplatinate reverses cyanide-induced metabolic derangements and counteracts severe cyanide poisoning.FASEB Bioadv. 2019 Feb;1(2):81-92. doi: 10.1096/fba.1024. Epub 2018 Oct 8. FASEB Bioadv. 2019. PMID: 31355359 Free PMC article.
-
Glyoxylate protects against cyanide toxicity through metabolic modulation.Sci Rep. 2022 Mar 23;12(1):4982. doi: 10.1038/s41598-022-08803-y. Sci Rep. 2022. PMID: 35322094 Free PMC article.
References
-
- Alcorta R. Smoke inhalation & acute cyanide poisoning. Hydrogen cyanide poisoning proves increasingly common in smoke-inhalation victims. JEMS. 2004;29(6-15) quiz suppl 16-17. - PubMed
-
- Anderson ME, Naganuma A, Meister A. Protection against cisplatin toxicity by administration of glutathione ester. FASEB J. 1990;4:3251–3255. - PubMed
-
- Banerjea D, Basolo F, Pearson RG. Mechanism of Substitution Reactions of Complex Ions. XII. Reactions of Some Platinum(II) Complexes with Various Reactants. Journal of the American Chemical Society. 1957;79:4055–4062.
-
- Barillo DJ, Goode R, Esch V. Cyanide poisoning in victims of fire: analysis of 364 cases and review of the literature. J Burn Care Rehabil. 1994;15:46–57. - PubMed
-
- Basolo F, Pearson RG. Progress in Inorganic Chemistry. John Wiley & Sons, Inc.; 2007. The Trans Effect in Metal Complexes; pp. 381–453.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

