Drebrin restricts rotavirus entry by inhibiting dynamin-mediated endocytosis
- PMID: 28416666
- PMCID: PMC5422808
- DOI: 10.1073/pnas.1619266114
Drebrin restricts rotavirus entry by inhibiting dynamin-mediated endocytosis
Abstract
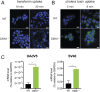
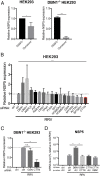
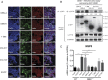
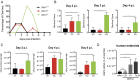
Despite the wide administration of several effective vaccines, rotavirus (RV) remains the single most important etiological agent of severe diarrhea in infants and young children worldwide, with an annual mortality of over 200,000 people. RV attachment and internalization into target cells is mediated by its outer capsid protein VP4. To better understand the molecular details of RV entry, we performed tandem affinity purification coupled with high-resolution mass spectrometry to map the host proteins that interact with VP4. We identified an actin-binding protein, drebrin (DBN1), that coprecipitates and colocalizes with VP4 during RV infection. Importantly, blocking DBN1 function by siRNA silencing, CRISPR knockout (KO), or chemical inhibition significantly increased host cell susceptibility to RV infection. Dbn1 KO mice exhibited higher incidence of diarrhea and more viral antigen shedding in their stool samples compared with the wild-type littermates. In addition, we found that uptake of other dynamin-dependent cargos, including transferrin, cholera toxin, and multiple viruses, was also enhanced in DBN1-deficient cells. Inhibition of cortactin or dynamin-2 abrogated the increased virus entry observed in DBN1-deficient cells, suggesting that DBN1 suppresses dynamin-mediated endocytosis via interaction with cortactin. Our study unveiled an unexpected role of DBN1 in restricting the entry of RV and other viruses into host cells and more broadly to function as a crucial negative regulator of diverse dynamin-dependent endocytic pathways.
Keywords: drebrin; endocytosis; rotavirus.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Rotavirus NSP1 Contributes to Intestinal Viral Replication, Pathogenesis, and Transmission.mBio. 2021 Dec 21;12(6):e0320821. doi: 10.1128/mBio.03208-21. Epub 2021 Dec 14. mBio. 2021. PMID: 34903043 Free PMC article.
-
Dynamin independent endocytosis is an alternative cell entry mechanism for multiple animal viruses.PLoS Pathog. 2024 Nov 14;20(11):e1012690. doi: 10.1371/journal.ppat.1012690. eCollection 2024 Nov. PLoS Pathog. 2024. PMID: 39541404 Free PMC article.
-
Rotaviruses reach late endosomes and require the cation-dependent mannose-6-phosphate receptor and the activity of cathepsin proteases to enter the cell.J Virol. 2014 Apr;88(8):4389-402. doi: 10.1128/JVI.03457-13. Epub 2014 Feb 5. J Virol. 2014. PMID: 24501398 Free PMC article.
-
Rotavirus Replication: Gaps of Knowledge on Virus Entry and Morphogenesis.Tohoku J Exp Med. 2019 Aug;248(4):285-296. doi: 10.1620/tjem.248.285. Tohoku J Exp Med. 2019. PMID: 31447474 Review.
-
[Research progress in receptors involved in rotavirus infection].Bing Du Xue Bao. 2014 May;30(3):303-9. Bing Du Xue Bao. 2014. PMID: 25118387 Review. Chinese.
Cited by
-
The Mettl3 epitranscriptomic writer amplifies p53 stress responses.Mol Cell. 2022 Jul 7;82(13):2370-2384.e10. doi: 10.1016/j.molcel.2022.04.010. Epub 2022 May 4. Mol Cell. 2022. PMID: 35512709 Free PMC article.
-
Rotavirus VP3 targets MAVS for degradation to inhibit type III interferon expression in intestinal epithelial cells.Elife. 2018 Nov 21;7:e39494. doi: 10.7554/eLife.39494. Elife. 2018. PMID: 30460894 Free PMC article.
-
The human phosphatase CDC14A modulates primary cilium length by regulating centrosomal actin nucleation.EMBO Rep. 2019 Jan;20(1):e46544. doi: 10.15252/embr.201846544. Epub 2018 Nov 22. EMBO Rep. 2019. PMID: 30467237 Free PMC article.
-
Screening and identification of cyprinid herpesvirus 2 (CyHV-2) ORF55-interacting proteins by phage display.Virol J. 2023 Apr 12;20(1):66. doi: 10.1186/s12985-023-02026-x. Virol J. 2023. PMID: 37046316 Free PMC article.
-
Drebrin 1 in dendritic cells regulates phagocytosis and cell surface receptor expression through recycling for efficient antigen presentation.Immunology. 2019 Feb;156(2):136-146. doi: 10.1111/imm.13010. Epub 2018 Nov 8. Immunology. 2019. PMID: 30317558 Free PMC article.
References
-
- Marsh M, editor. 2001. Endocytosis, Frontiers in Molecular Biology (Oxford University Press, Oxford)
-
- McMahon HT, Boucrot E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat Rev Mol Cell Biol. 2011;12:517–533. - PubMed
-
- Mercer J, Schelhaas M, Helenius A. Virus entry by endocytosis. Annu Rev Biochem. 2010;79:803–833. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

