Enantioselective Heck-Matsuda Arylations through Chiral Anion Phase-Transfer of Aryl Diazonium Salts
- PMID: 28418118
- PMCID: PMC5528849
- DOI: 10.1002/anie.201702107
Enantioselective Heck-Matsuda Arylations through Chiral Anion Phase-Transfer of Aryl Diazonium Salts
Abstract
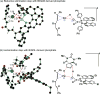
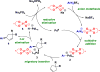
A mild, asymmetric Heck-Matsuda reaction of five-, six- and seven-membered ring alkenes and aryl diazonium salts is presented. High yields and enantioselectivities were achieved using Pd0 and chiral anion co-catalysts, the latter functioning as a chiral anion phase-transfer (CAPT) reagent. For certain substrate classes, the chiral anion catalysts were modulated to minimize the formation of undesired by-products. More specifically, BINAM-derived phosphoric acid catalysts were shown to prevent alkene isomerization in cyclopentene and cycloheptene starting materials. DFT(B3LYP-D3) computations revealed that increased product selectivity resulted from a chiral anion dependent lowering of the activation barrier for the desired pathway.
Keywords: Heck reaction; Heck-Matsuda reaction; chiral anions; palladium; phase-transfer catalysis.
© 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

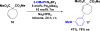

Similar articles
-
Scope and limitations of the Heck-Matsuda-coupling of phenol diazonium salts and styrenes: a protecting-group economic synthesis of phenolic stilbenes.Org Biomol Chem. 2013 Jun 14;11(22):3674-91. doi: 10.1039/c3ob40420j. Org Biomol Chem. 2013. PMID: 23615777
-
Enantioselective Heck arylations of acyclic alkenyl alcohols using a redox-relay strategy.Science. 2012 Dec 14;338(6113):1455-8. doi: 10.1126/science.1229208. Science. 2012. PMID: 23239733 Free PMC article.
-
Pd-catalyzed arylation reactions with phenol diazonium salts: application in the synthesis of diarylheptanoids.J Org Chem. 2011 May 6;76(9):3357-65. doi: 10.1021/jo2002787. Epub 2011 Apr 6. J Org Chem. 2011. PMID: 21434690
-
From α-arylation of olefins to acylation with aldehydes: a journey in regiocontrol of the Heck reaction.Acc Chem Res. 2011 Aug 16;44(8):614-26. doi: 10.1021/ar200053d. Epub 2011 May 25. Acc Chem Res. 2011. PMID: 21612205 Review.
-
Enantioselective Dicarbofunctionalization of Unactivated Alkenes by Palladium-Catalyzed Tandem Heck/Suzuki Coupling Reaction.Angew Chem Int Ed Engl. 2019 Oct 7;58(41):14653-14659. doi: 10.1002/anie.201907840. Epub 2019 Sep 5. Angew Chem Int Ed Engl. 2019. PMID: 31420928 Review.
Cited by
-
New anionic cobalt(III) complexes enable enantioselective synthesis of spiro-fused oxazoline and iodoacetal derivatives.Front Chem. 2022 Oct 13;10:1034291. doi: 10.3389/fchem.2022.1034291. eCollection 2022. Front Chem. 2022. PMID: 36311431 Free PMC article.
-
Modular preparation of cationic bipyridines and azaarenes via C-H activation.Chem Sci. 2023 Nov 15;14(46):13530-13536. doi: 10.1039/d3sc04864k. eCollection 2023 Nov 29. Chem Sci. 2023. PMID: 38033896 Free PMC article.
-
Origin of stereoselectivity in the amination of alcohols using cooperative asymmetric dual catalysis involving chiral counter-ions.Chem Sci. 2018 Jun 25;9(28):6126-6133. doi: 10.1039/c8sc01433g. eCollection 2018 Jul 28. Chem Sci. 2018. PMID: 30090300 Free PMC article.
-
Atroposelective Synthesis of 2,2'-Bis(arylamino)-1,1'-biaryls by Oxidative Iron(III)- and Phosphoric Acid-Catalyzed C-C Coupling of Diarylamines.Chemistry. 2023 Jan 27;29(6):e202203269. doi: 10.1002/chem.202203269. Epub 2022 Nov 27. Chemistry. 2023. PMID: 36269611 Free PMC article.
-
Enantioselective Intramolecular Allylic Substitution via Synergistic Palladium/Chiral Phosphoric Acid Catalysis: Insight into Stereoinduction through Statistical Modeling.Angew Chem Int Ed Engl. 2020 Aug 17;59(34):14647-14655. doi: 10.1002/anie.202006237. Epub 2020 Jun 30. Angew Chem Int Ed Engl. 2020. PMID: 32453890 Free PMC article.
References
-
- Kikukawa K, Matsuda T. Chem Lett. 1977;6:159–162.
-
- Roglans A, Pla-Quintana A, Moreno-Mañas M. Chem Rev. 2006;106:4622–4643. - PubMed
- Taylor JG, Moro AV, Correia CRD. Eur J Org Chem. 2011;2011:1403–1428.
- Mo F, Dong G, Zhang Y, Wang J. Org Biomol Chem. 2013;11:1582–1593. - PubMed
- Felpin FX, Nassar-Hardy L, Le Callonnec F, Fouquet E. Tetrahedron. 2011;67:2815–2831.
-
- Balz G, Schiemann G. Ber dtsch Chem Ges A/B. 1927;60:1186–1190.
-
- Andrus MB, Song C. Org Lett. 2001;3:3761–3764. - PubMed
- Kikukawa K, Nagira K, Wada F, Matsuda T. Tetrahedron. 1981;37:31–36.
- Sengupta S, Bhattacharya S. J Chem Soc, Perkin Trans 1. 1993:1943–1944.
-
- Dai M, Liang B, Wang C, Chen J, Yang Z. Org Lett. 2004;6:221–224. - PubMed
- Masllorens J, Moreno-Mañas M, Pla-Quintana A, Roglans A. Org Lett. 2003;5:1559–1561. - PubMed
- Brunner H, de Courcy NLC, Genêt J-P. Tetrahedron Lett. 1999;40:4815–4818.
- Masllorens J, Bouquillon S, Roglans A, Hénin F, Muzart J. J Organomet Chem. 2005;690:3822–3826.
- Andrus MB, Song C, Zhang J. Org Lett. 2002;4:2079–2082. - PubMed
- Severino EA, Correia CRD. Org Lett. 2000;2:3039–3042. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

