Roles of long noncoding RNAs in colorectal cancer metastasis
- PMID: 28418892
- PMCID: PMC5503659
- DOI: 10.18632/oncotarget.16339
Roles of long noncoding RNAs in colorectal cancer metastasis
Abstract
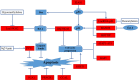
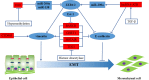
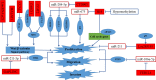
Colorectal cancer (CRC) is the 3rd most common malignancies worldwide. Metastasis is responsible for more than 90% CRC patients' death. Long noncoding RNAs (lncRNAs) are an important class of transcribed RNA molecules greater than 200 nucleotides in length. With the development of whole genome sequencing technologies, they have been gained more attention. Accumulating evidences suggest that abnormal expression of lncRNAs in diverse diseases are involved in various biological functions such as proliferation, apoptosis, metastasis and differentiation by acting as epigenetic, splicing, transcriptional or post-transcriptional regulators. Aberrant expression of lncRNAs has also been found in CRC. Besides, recent studies have indicated that lncRNAs play important roles in tumourigenesis and cancer metastasis. They participate in the process of metastasis by activing or inhibiting the metastatic pathways. However, their functions on the development of cancer metastasis are poorly understood. In this review, we highlight the findings of roles for lncRNAs in CRC metastasis and review the metastatic pathways of lncRNAs leading to cancer metastasis in CRC, including escape of apoptosis, epithelial-mesenchymal transition (EMT), angiogenesis and invasion, migration and proliferation. Furthermore, we also discuss the potential clinical application of lncRNAs in CRC as diagnostic markers and therapeutic targets.
Keywords: colorectal cancer; lncRNAs; metastasis; review.
Conflict of interest statement
All authors declare no conflict of interest. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Figures
Similar articles
-
LncRNAs: the bridge linking RNA and colorectal cancer.Oncotarget. 2017 Feb 14;8(7):12517-12532. doi: 10.18632/oncotarget.13573. Oncotarget. 2017. PMID: 27888635 Free PMC article. Review.
-
Long noncoding RNAs: novel players in colorectal cancer.Cancer Lett. 2015 May 28;361(1):13-21. doi: 10.1016/j.canlet.2015.03.002. Epub 2015 Mar 6. Cancer Lett. 2015. PMID: 25754818 Review.
-
Long non-coding RNAs: a rising biotarget in colorectal cancer.Oncotarget. 2017 Mar 28;8(13):22187-22202. doi: 10.18632/oncotarget.14728. Oncotarget. 2017. PMID: 28108736 Free PMC article. Review.
-
The Biological Effect and Clinical Application of Long Noncoding RNAs in Colorectal Cancer.Cell Physiol Biochem. 2018;46(2):431-441. doi: 10.1159/000488610. Epub 2018 Mar 26. Cell Physiol Biochem. 2018. PMID: 29614491 Review.
-
Interactions among lncRNAs, miRNAs and mRNA in colorectal cancer.Biochimie. 2019 Aug;163:58-72. doi: 10.1016/j.biochi.2019.05.010. Epub 2019 May 10. Biochimie. 2019. PMID: 31082429 Review.
Cited by
-
[Long non-coding RNA LINC01106 regulates colorectal cancer cell proliferation and apoptosis through the STAT3 pathway].Nan Fang Yi Ke Da Xue Xue Bao. 2020 Sep 30;40(9):1259-1264. doi: 10.12122/j.issn.1673-4254.2020.09.06. Nan Fang Yi Ke Da Xue Xue Bao. 2020. PMID: 32990221 Free PMC article. Chinese.
-
Role of Curcumin in Regulating Long Noncoding RNA Expression in Cancer.Adv Exp Med Biol. 2021;1308:13-23. doi: 10.1007/978-3-030-64872-5_2. Adv Exp Med Biol. 2021. PMID: 33861433
-
Screening and functional analysis of differentially expressed lncRNAs in rapid atrial pacing dog atrial tissue.J Interv Card Electrophysiol. 2021 Aug;61(2):375-384. doi: 10.1007/s10840-020-00824-9. Epub 2020 Jul 15. J Interv Card Electrophysiol. 2021. PMID: 32671717
-
POFUT1 promotes colorectal cancer development through the activation of Notch1 signaling.Cell Death Dis. 2018 Sep 24;9(10):995. doi: 10.1038/s41419-018-1055-2. Cell Death Dis. 2018. PMID: 30250219 Free PMC article.
-
Long noncoding RNA MALAT1 regulates HDAC4-mediated proliferation and apoptosis via decoying of miR-140-5p in osteosarcoma cells.Cancer Med. 2018 Sep;7(9):4584-4597. doi: 10.1002/cam4.1677. Epub 2018 Aug 9. Cancer Med. 2018. PMID: 30094957 Free PMC article.
References
-
- Schmidt-Kittler O, Ragg T, Daskalakis A, Granzow M, Ahr A, Blankenstein TJ, Kaufmann M, Diebold J, Arnholdt H, Muller P, Bischoff J, Harich D, Schlimok G, et al. From latent disseminated cells to overt metastasis: genetic analysis of systemic breast cancer progression. Proc Natl Acad Sci U S A. 2003;100:7737–42. doi: 10.1073/pnas.1331931100. - DOI - PMC - PubMed
-
- Stein U, Schlag PM. Clinical, biological, and molecular aspects of metastasis in colorectal cancer. Recent Results Cancer Res. 2007;176:61–80. - PubMed
-
- Grothey A, Schmoll HJ. New chemotherapy approaches in colorectal cancer. Curr Opin Oncol. 2001;13:275–86. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

