Antimicrobial peptides with selective antitumor mechanisms: prospect for anticancer applications
- PMID: 28422728
- PMCID: PMC5542299
- DOI: 10.18632/oncotarget.16743
Antimicrobial peptides with selective antitumor mechanisms: prospect for anticancer applications
Abstract
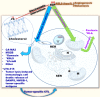
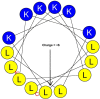
In the last several decades, there have been significant advances in anticancer therapy. However, the development of resistance to cancer drugs and the lack of specificity related to actively dividing cells leading to toxic side effects have undermined these achievements. As a result, there is considerable interest in alternative drugs with novel antitumor mechanisms. In addition to the recent approach using immunotherapy, an effective but much cheaper therapeutic option of pharmaceutical drugs would still provide the best choice for cancer patients as the first line treatment. Ribosomally synthesized cationic antimicrobial peptides (AMPs) or host defense peptides (HDP) display broad-spectrum activity against bacteria based on electrostatic interactions with negatively charged lipids on the bacterial surface. Because of increased proportions of phosphatidylserine (negatively charged) on the surface of cancer cells compared to normal cells, cationic amphipathic peptides could be an effective source of anticancer agents that are both selective and refractory to current resistance mechanisms. We reviewed herein the prospect for AMP application to cancer treatment, with a focus on modes of action of cationic AMPs.
Keywords: anticancer peptides; antimicrobial peptides; antitumor peptides; cationic peptides; host defense peptides.
Conflict of interest statement
None.
Figures


References
-
- Hashim D, Boffetta P, La Vecchia C, Rota M, Bertuccio P, Malvezzi M, Negri E. The global decrease in cancer mortality: trends and disparities. Ann Oncol. 2016;27:926–933. - PubMed
-
- Torre LA, Siegel RL, Ward EM, Jemal A. Global Cancer Incidence and Mortality Rates and Trends–An Update. Cancer Epidemiol Biomarkers Prev. 2016;25:16–27. - PubMed
-
- Saika K, Machii R. Cancer mortality attributable to tobacco by region based on the WHO Global Report. Jpn J Clin Oncol. 2012;42:771–72. - PubMed
-
- Daskivich TJ. Life Expectancy and Treatment Choice for Men with High-risk Prostate Cancer. Eur Urol. 2015;68:59–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

