Two year result of intravitreal bevacizumab for diabetic macular edema using treat and extend protocol
- PMID: 28422832
- PMCID: PMC5406048
- DOI: 10.1097/MD.0000000000006406
Two year result of intravitreal bevacizumab for diabetic macular edema using treat and extend protocol
Abstract
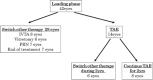
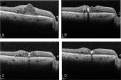
To determine the efficacy of the treat and extend (TAE) protocol with intravitreal bevacizumab (IVB) for managing diabetic macular edema (DME).Retrospective, single-center study.For this retrospective study, 42 eyes of 42 patients were initially treated with 3 consecutive monthly IVB injections (loading phase), after which they were selected for different additional therapies. For the TAE protocol, the baseline treatment interval was selected to be 8 weeks and was sequentially lengthened by 2 weeks if the central macular thickness (CMT) was <300 μm at 2 consecutive examinations.Among the 42 eyes, 8 eyes (19.0%) received the TAE treatment for 2 years. The BCVA was improved significantly from 0.37 ± 0.04 before treatment to 0.19 ± 0.04 logMAR units at 2 years after the TAE determined IVB injections (P < .05). The ratio of eyes with a gain of the BCVA by more than 2 lines was 37.5%. The CMT was significantly reduced from 515.4 ± 75.5 to 303.6 ± 45.0 μm after 2 years (P < .01). The mean number of TAE injection was 8.8 and the mean injection interval was 11.0 weeks.After the loading phase, 19.0% of patients can be treated with the TAE protocol. Although significant visual improvements were obtained after the TAE protocol, it does not apply to every DME case.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures
Similar articles
-
Intravitreal Diclofenac plus Bevacizumab versus Bevacizumab alone in treatment-naive diabetic macular edema: a randomized double-blind clinical trial.Int Ophthalmol. 2017 Aug;37(4):867-874. doi: 10.1007/s10792-016-0335-z. Epub 2016 Sep 13. Int Ophthalmol. 2017. PMID: 27624173 Clinical Trial.
-
Intravitreal bevacizumab and dexamethasone implant for treatment of chronic diabetic macular edema.Cutan Ocul Toxicol. 2017 Jun;36(2):180-184. doi: 10.3109/15569527.2015.1127254. Epub 2017 Mar 1. Cutan Ocul Toxicol. 2017. PMID: 28366074 Clinical Trial.
-
Intravitreal bevacizumab and/or macular photocoagulation as a primary treatment for diffuse diabetic macular edema.Retina. 2010 Nov-Dec;30(10):1638-45. doi: 10.1097/IAE.0b013e3181e1ed07. Retina. 2010. PMID: 20838357 Clinical Trial.
-
Intravitreal bevacizumab for diabetic macular oedema: 5-year results of the Pan-American Collaborative Retina Study group.Br J Ophthalmol. 2016 Dec;100(12):1605-1610. doi: 10.1136/bjophthalmol-2015-307950. Epub 2016 Feb 24. Br J Ophthalmol. 2016. PMID: 26912377
-
Primary intravitreal bevacizumab for diffuse diabetic macular edema: the Pan-American Collaborative Retina Study Group at 24 months.Ophthalmology. 2009 Aug;116(8):1488-97, 1497.e1. doi: 10.1016/j.ophtha.2009.03.016. Epub 2009 Jul 9. Ophthalmology. 2009. PMID: 19545900
Cited by
-
An anchoring molecule increases intravitreal retention of antibody-based therapeutics used in the treatment of ocular diseases.J Control Release. 2020 Dec 10;328:263-275. doi: 10.1016/j.jconrel.2020.08.034. Epub 2020 Aug 25. J Control Release. 2020. PMID: 32858075 Free PMC article.
-
One-Year Outcomes of Aflibercept in Treat-and-Extend Versus Pro Re Nata Regimens for Bevacizumab-Resistant Diabetic Macular Edema: A Real-World Study.Ophthalmol Ther. 2025 Jan;14(1):169-181. doi: 10.1007/s40123-024-01067-x. Epub 2024 Nov 22. Ophthalmol Ther. 2025. PMID: 39576487 Free PMC article.
-
Real-World Evidence in the Management of Diabetic Macular Edema with Intravitreal Anti-VEGFs in Asia: A Systematic Literature Review.Clin Ophthalmol. 2022 Oct 19;16:3503-3526. doi: 10.2147/OPTH.S378392. eCollection 2022. Clin Ophthalmol. 2022. PMID: 36274678 Free PMC article. Review.
-
Glucose-regulated protein 78 in the aqueous humor in diabetic macular edema patients.Medicine (Baltimore). 2018 Nov;97(45):e12757. doi: 10.1097/MD.0000000000012757. Medicine (Baltimore). 2018. PMID: 30407281 Free PMC article.
-
Real-World Use of Off-Label MVASI in the Treatment of Patients With Neovascular AMD and DME.Transl Vis Sci Technol. 2024 Jul 1;13(7):17. doi: 10.1167/tvst.13.7.17. Transl Vis Sci Technol. 2024. PMID: 39042047 Free PMC article.
References
-
- Moss SE, Klein R, Klein BEK. The incidence of visual loss in a diabetic population. Ophthalmology 1988;95:1340–8. - PubMed
-
- McMeel JW, Trempe CL, Franks EB. Diabetic maculopathy. Trans Sect Ophthalmol Am Acad Ophthalmol Otolaryngol 1977;83:476–87. - PubMed
-
- Arevalo JF, Fromow-Guerra J, et al. Primary intravitreal bevacizumab (Avastin) for diabetic macular edema. Ophthalmology 2007;114:743–50. - PubMed
-
- Haritoglou C, Kook D, Neubauer A, et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina 2006;26:999–1005. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

