Whole-Exome Sequencing of Metaplastic Breast Carcinoma Indicates Monoclonality with Associated Ductal Carcinoma Component
- PMID: 28424200
- PMCID: PMC5559334
- DOI: 10.1158/1078-0432.CCR-17-0108
Whole-Exome Sequencing of Metaplastic Breast Carcinoma Indicates Monoclonality with Associated Ductal Carcinoma Component
Abstract
Purpose: Although most human cancers display a single histology, there are unusual cases where two or more distinct tissue types present within a primary tumor. One such example is metaplastic breast carcinoma, a rare but aggressive cancer with a heterogeneous histology, including squamous, chondroid, and spindle cells. Metaplastic carcinomas often contain an admixed conventional ductal invasive or in situ mammary carcinoma component, and are typically triple-negative for estrogen receptor, progesterone receptor, and HER-2 amplification/overexpression. An unanswered question is the origin of metaplastic breast cancers. While they may arise independently from their ductal components, their close juxtaposition favors a model that postulates a shared origin, either as two derivatives from the same primary cancer or one histology as an outgrowth of the other. Understanding the mechanism of development of these tumors may inform clinical decisions.Experimental Design: We performed exome sequencing for paired metaplastic and adjacent conventional invasive ductal carcinomas in 8 patients and created a pipeline to identify somatic variants and predict their functional impact, without having normal tissue. We then determined the genetic relationships between the histologically distinct compartments.Results: In each case, the tumor components have nearly identical landscapes of somatic mutation, implying that the differing histologies do not derive from genetic clonal divergence.Conclusions: A shared origin for tumors with differing histologies suggests that epigenetic or noncoding changes may mediate the metaplastic phenotype and that alternative therapeutic approaches, including epigenetic therapies, may be required for metaplastic breast cancers. Clin Cancer Res; 23(16); 4875-84. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures


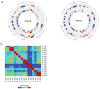
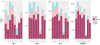


References
-
- Reis-Filho JSLS, Gobbi H, Sneige N. Metaplastic carcinoma. In: Lakhani SREI, schnitt SJ, tan PH, van de vijver MJ, editors. world health organization classification of tumours of the breast. 2012. p. 48.
-
- Wargotz ES, Deos PH, Norris HJ. Metaplastic carcinomas of the breast. II. spindle cell carcinoma. Hum Pathol. 1989;20(8):732–40. - PubMed
-
- Wargotz ES, Norris HJ. Metaplastic carcinomas of the breast. IV. squamous cell carcinoma of ductal origin. Cancer. 1990;65(2):272–6. - PubMed
-
- Wargotz ES, Norris HJ. Metaplastic carcinomas of the breast. III. carcinosarcoma. Cancer. 1989;64(7):1490–9. - PubMed
-
- Wargotz ES, Norris HJ. Metaplastic carcinomas of the breast. I. matrix-producing carcinoma. Hum Pathol. 1989;20(7):628–35. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

