Tension-activated channels in the mechanism of osmotic fitness in Pseudomonas aeruginosa
- PMID: 28424229
- PMCID: PMC5412531
- DOI: 10.1085/jgp.201611699
Tension-activated channels in the mechanism of osmotic fitness in Pseudomonas aeruginosa
Abstract
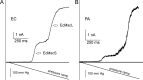
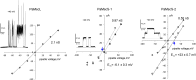
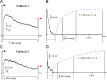
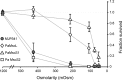
Pseudomonas aeruginosa (PA) is an opportunistic pathogen with an exceptional ability to adapt to a range of environments. Part of its adaptive potential is the ability to survive drastic osmolarity changes. Upon a sudden dilution of external medium, such as during exposure to rain, bacteria evade mechanical rupture by engaging tension-activated channels that act as osmolyte release valves. In this study, we compare fast osmotic permeability responses in suspensions of wild-type PA and Escherichia coli (EC) strains in stopped-flow experiments and provide electrophysiological descriptions of osmotic-release channels in PA. Using osmotic dilution experiments, we first show that PA tolerates a broader range of shocks than EC. We record the kinetics of cell equilibration reported by light scattering responses to osmotic up- and down-shocks. PA exhibits a lower water permeability and faster osmolyte release rates during large osmotic dilutions than EC, which correlates with better survival. To directly characterize the PA tension-activated channels, we generate giant spheroplasts from this microorganism and record current responses in excised patches. Unlike EC, which relies primarily on two types of channels, EcMscS and EcMscL, to generate a distinctive two-wave pressure ramp response, PA exhibits a more gradual response that is dominated by MscL-type channels. Genome analysis, cloning, and expression reveal that PA possesses one MscL-type (PaMscL) and two MscS-type (PaMscS-1 and 2) proteins. In EC spheroplasts, both PaMscS channels exhibit a slightly earlier activation by pressure compared with EcMscS. Unitary currents reveal that PaMscS-2 has a smaller conductance, higher anionic preference, stronger inactivation, and slower recovery compared with PaMscS-1. We conclude that PA relies on MscL as the major valve defining a high rate of osmolyte release sufficient to curb osmotic swelling under extreme shocks, but it still requires MscS-type channels with a strong propensity to inactivation to properly terminate massive permeability response.
© 2017 Çetiner et al.
Figures





Comment in
-
Pseudomonas doesn't mind a dunking.J Gen Physiol. 2017 May 1;149(5):531. doi: 10.1085/jgp.201711799. Epub 2017 Apr 19. J Gen Physiol. 2017. PMID: 28424228 Free PMC article.
Similar articles
-
Characterizing the mechanosensitive response of Paraburkholderia graminis membranes.Biochim Biophys Acta Biomembr. 2020 Apr 1;1862(4):183176. doi: 10.1016/j.bbamem.2020.183176. Epub 2020 Jan 7. Biochim Biophys Acta Biomembr. 2020. PMID: 31923411
-
Mechanosensitive channel MscS is critical for termination of the bacterial hypoosmotic permeability response.J Gen Physiol. 2023 May 1;155(5):e202213168. doi: 10.1085/jgp.202213168. Epub 2023 Apr 6. J Gen Physiol. 2023. PMID: 37022337 Free PMC article.
-
Mechanosensitive channel MscS is critical for termination of the bacterial hypoosmotic permeability response.bioRxiv [Preprint]. 2023 Mar 1:2023.02.27.530336. doi: 10.1101/2023.02.27.530336. bioRxiv. 2023. Update in: J Gen Physiol. 2023 May 1;155(5):e202213168. doi: 10.1085/jgp.202213168. PMID: 36909569 Free PMC article. Updated. Preprint.
-
The MscS and MscL families of mechanosensitive channels act as microbial emergency release valves.J Bacteriol. 2012 Sep;194(18):4802-9. doi: 10.1128/JB.00576-12. Epub 2012 Jun 8. J Bacteriol. 2012. PMID: 22685280 Free PMC article. Review.
-
From membrane tension to channel gating: A principal energy transfer mechanism for mechanosensitive channels.Protein Sci. 2016 Nov;25(11):1954-1964. doi: 10.1002/pro.3017. Epub 2016 Aug 23. Protein Sci. 2016. PMID: 27530280 Free PMC article. Review.
Cited by
-
The voltage-dependence of MscL has dipolar and dielectric contributions and is governed by local intramembrane electric field.Sci Rep. 2018 Sep 11;8(1):13607. doi: 10.1038/s41598-018-31945-x. Sci Rep. 2018. PMID: 30206263 Free PMC article.
-
Bacterial and fungal biodeterioration of discolored building paints in Lagos, Nigeria.World J Microbiol Biotechnol. 2017 Oct 5;33(11):196. doi: 10.1007/s11274-017-2362-y. World J Microbiol Biotechnol. 2017. PMID: 28983733
-
Tight hydrophobic core and flexible helices yield MscL with a high tension gating threshold and a membrane area mechanical strain buffer.Front Chem. 2023 May 24;11:1159032. doi: 10.3389/fchem.2023.1159032. eCollection 2023. Front Chem. 2023. PMID: 37292176 Free PMC article.
-
Spatiotemporal relationships defining the adaptive gating of the bacterial mechanosensitive channel MscS.Eur Biophys J. 2018 Sep;47(6):663-677. doi: 10.1007/s00249-018-1303-5. Epub 2018 Apr 23. Eur Biophys J. 2018. PMID: 29687344
-
In vitro antibacterial activity of bioactive glass S53P4 on multiresistant pathogens causing osteomyelitis and prosthetic joint infection.BMC Infect Dis. 2018 Apr 3;18(1):157. doi: 10.1186/s12879-018-3069-x. BMC Infect Dis. 2018. PMID: 29614973 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

