Janus Kinase 2 Regulates Transcription Factor EB Expression and Autophagy Completion in Glomerular Podocytes
- PMID: 28424277
- PMCID: PMC5576936
- DOI: 10.1681/ASN.2016111208
Janus Kinase 2 Regulates Transcription Factor EB Expression and Autophagy Completion in Glomerular Podocytes
Abstract
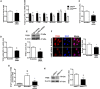
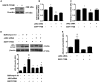
The nonreceptor kinase Janus kinase 2 (JAK2) has garnered attention as a promising therapeutic target for the treatment of CKD. However, being ubiquitously expressed in the adult, JAK2 is also likely to be necessary for normal organ function. Here, we investigated the phenotypic effects of JAK2 deficiency. Mice in which JAK2 had been deleted from podocytes exhibited an elevation in urine albumin excretion that was accompanied by increased podocyte autophagosome fractional volume and p62 aggregation, which are indicative of impaired autophagy completion. In cultured podocytes, knockdown of JAK2 similarly impaired autophagy and led to downregulation in the expression of lysosomal genes and decreased activity of the lysosomal enzyme, cathepsin D. Because transcription factor EB (TFEB) has recently emerged as a master regulator of autophagosome-lysosome function, controlling the expression of several of the genes downregulated by JAK2 knockdown, we questioned whether TFEB is regulated by JAK2. In immortalized mouse podocytes, JAK2 knockdown decreased TFEB promoter activity, expression, and nuclear localization. In silico analysis and chromatin immunoprecipitation assays revealed that the downstream mediator of JAK2 signaling STAT1 binds to the TFEB promoter. Finally, overexpression of TFEB in JAK2-deficient podocytes reversed lysosomal dysfunction and restored albumin permselectivity. Collectively, these observations highlight the homeostatic actions of JAK2 in podocytes and the importance of TFEB to autophagosome-lysosome function in these cells. These results also raise the possibility that therapeutically modulating TFEB activity may improve podocyte health in glomerular disease.
Keywords: JAK2; autophagy; lysosome; podocyte.
Copyright © 2017 by the American Society of Nephrology.
Figures



References
-
- Hartleben B, Gödel M, Meyer-Schwesinger C, Liu S, Ulrich T, Köbler S, Wiech T, Grahammer F, Arnold SJ, Lindenmeyer MT, Cohen CD, Pavenstädt H, Kerjaschki D, Mizushima N, Shaw AS, Walz G, Huber TB: Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. J Clin Invest 120: 1084–1096, 2010 - PMC - PubMed
-
- Logar CM, Brinkkoetter PT, Krofft RD, Pippin JW, Shankland SJ: Darbepoetin alfa protects podocytes from apoptosis in vitro and in vivo. Kidney Int 72: 489–498, 2007 - PubMed
-
- Chuang PY, He JC: JAK/STAT signaling in renal diseases. Kidney Int 78: 231–234, 2010 - PubMed
-
- Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR, Tichelli A, Cazzola M, Skoda RC: A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 352: 1779–1790, 2005 - PubMed
-
- Berthier CC, Zhang H, Schin M, Henger A, Nelson RG, Yee B, Boucherot A, Neusser MA, Cohen CD, Carter-Su C, Argetsinger LS, Rastaldi MP, Brosius FC, Kretzler M: Enhanced expression of Janus kinase-signal transducer and activator of transcription pathway members in human diabetic nephropathy. Diabetes 58: 469–477, 2009 - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

