A lack of coordination between sister-chromatids segregation and cytokinesis in the oocytes of B6.YTIR (XY) sex-reversed female mice
- PMID: 28424461
- PMCID: PMC5430445
- DOI: 10.1038/s41598-017-00922-1
A lack of coordination between sister-chromatids segregation and cytokinesis in the oocytes of B6.YTIR (XY) sex-reversed female mice
Abstract
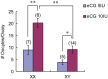
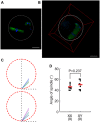
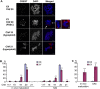
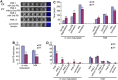
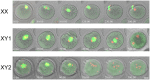
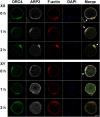
The B6.YTIR (XY) mouse develops bilateral ovaries despite the expression of the testis-determining gene Sry during gonadal differentiation. We reported that the oocytes of the XY female are defective in their cytoplasm, resulting in a failure in the second meiotic division after activation or fertilization in vitro. However, the mechanism of meiotic failure or the cause of infertility remained to be clarified. In the present study, we obtained mature oocytes from XY females by superovulation and confirmed that these oocytes also fail in zygotic development. By using confocal microscopy 3D-analysis, we demonstrated that meiotic spindles were properly positioned and oriented in the MII-oocytes from XY females. After parthenogenic activation, fewer oocytes from XY females extruded the second polar body, and in those oocytes, sister-chromatids were often separated but neither set entered the second polar body. ARP2, F-actin, and ORC4, known to play roles in asymmetric meiotic division, were initially localized along the ooplasmic membrane and concentrated over the MII-spindle but lost their cortical polarity after activation while the sister-chromatids moved away from the oolemma in the oocytes from XY females. Our results indicate that the second polar body extrusion is uncoupled from the sister-chromatids separation in the oocytes from XY female mouse.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
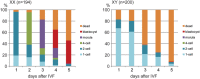
References
-
- Lovell-Badge R, Robertson E. XY female mice resulting from a heritable mutation in the primary testis-determining gene. Tdy. Development. 1990;109:635–646. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

