Hyperpolarized 13C Spectroscopic Evaluation of Oxidative Stress in a Rodent Model of Steatohepatitis
- PMID: 28425467
- PMCID: PMC5397869
- DOI: 10.1038/srep46014
Hyperpolarized 13C Spectroscopic Evaluation of Oxidative Stress in a Rodent Model of Steatohepatitis
Abstract
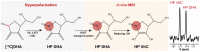
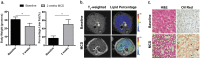
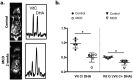
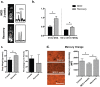
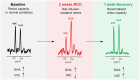
Nonalcoholic fatty liver disease (NAFLD) has become highly prevalent, now considered the most common liver disease in the western world. Approximately one-third of patients with NASH develop non-alchoholic steatohepatitis (NASH), histologically defined by lobular and portal inflammation, and accompanied by marked oxidative stress. Patients with NASH are at increased risk for cirrhosis and hepatocellular carcinoma, and diagnosis currently requires invasive biopsy. In animal models of NASH, particularly the methionine-choline deficient (MCD) model, profound changes are seen in redox enzymes and key intracellular antioxidants. To study antioxidant status in NASH non-invasively, we applied the redox probe hyperpolarized [1-13C] dehydroascorbic acid (HP DHA), which is reduced to Vitamin C (VitC) rapidly in the normal liver. In MCD mice, we observed a significant decrease in HP DHA to VitC conversion that accompanied hepatic fat deposition. When these animals were subsequently placed on a normal diet, resonance ratios reverted to those seen in control mice. These findings suggest that HP DHA, a potentially clinically translatable imaging agent, holds special promise in imaging NASH and other metabolic syndromes, to monitor disease progression and response to targeted therapies.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
Epigallocatechin gallate attenuated non-alcoholic steatohepatitis induced by methionine- and choline-deficient diet.Eur J Pharmacol. 2015 Aug 15;761:405-12. doi: 10.1016/j.ejphar.2015.05.005. Epub 2015 May 9. Eur J Pharmacol. 2015. PMID: 25967348
-
Alpha-syntrophin null mice are protected from non-alcoholic steatohepatitis in the methionine-choline-deficient diet model but not the atherogenic diet model.Biochim Biophys Acta Mol Cell Biol Lipids. 2018 May;1863(5):526-537. doi: 10.1016/j.bbalip.2018.02.006. Epub 2018 Feb 21. Biochim Biophys Acta Mol Cell Biol Lipids. 2018. PMID: 29474931
-
Chicoric acid attenuate a nonalcoholic steatohepatitis by inhibiting key regulators of lipid metabolism, fibrosis, oxidation, and inflammation in mice with methionine and choline deficiency.Mol Nutr Food Res. 2017 May;61(5). doi: 10.1002/mnfr.201600632. Epub 2017 Feb 22. Mol Nutr Food Res. 2017. PMID: 27981809
-
Necroptosis is a key pathogenic event in human and experimental murine models of non-alcoholic steatohepatitis.Clin Sci (Lond). 2015 Oct 1;129(8):721-39. doi: 10.1042/CS20140732. Epub 2015 Jun 15. Clin Sci (Lond). 2015. PMID: 26201023
-
Does vitamin C deficiency promote fatty liver disease development?Nutrients. 2014 Dec 1;6(12):5473-99. doi: 10.3390/nu6125473. Nutrients. 2014. PMID: 25533004 Free PMC article. Review.
Cited by
-
Detection of early-stage NASH using non-invasive hyperpolarized 13C metabolic imaging.Sci Rep. 2024 Jun 27;14(1):14854. doi: 10.1038/s41598-024-65951-z. Sci Rep. 2024. PMID: 38937567 Free PMC article.
-
Multi-nuclear magnetic resonance spectroscopy: state of the art and future directions.Insights Imaging. 2022 Aug 17;13(1):135. doi: 10.1186/s13244-022-01262-z. Insights Imaging. 2022. PMID: 35976510 Free PMC article. Review.
-
Detecting liver injury non-invasively using hyperpolarized 13 C MRI.Liver Int. 2018 Jun;38(6):988-990. doi: 10.1111/liv.13726. Liver Int. 2018. PMID: 29863314 Free PMC article. No abstract available.
-
Hyperpolarized 13 C magnetic resonance spectroscopy detects toxin-induced neuroinflammation in mice.NMR Biomed. 2019 Nov;32(11):e4164. doi: 10.1002/nbm.4164. Epub 2019 Aug 22. NMR Biomed. 2019. PMID: 31437326 Free PMC article.
-
Hyperpolarized Magnetic Resonance and Artificial Intelligence: Frontiers of Imaging in Pancreatic Cancer.JMIR Med Inform. 2021 Jun 17;9(6):e26601. doi: 10.2196/26601. JMIR Med Inform. 2021. PMID: 34137725 Free PMC article. Review.
References
-
- Chitturi S. & Farrell G. C. Etiopathogenesis of nonalcoholic steatohepatitis. Semin Liver Dis 21, 27–41 (2001). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

