Cabozantinib in hepatocellular carcinoma: results of a phase 2 placebo-controlled randomized discontinuation study
- PMID: 28426123
- PMCID: PMC5391701
- DOI: 10.1093/annonc/mdw651
Cabozantinib in hepatocellular carcinoma: results of a phase 2 placebo-controlled randomized discontinuation study
Abstract
Background: Cabozantinib, an orally bioavailable inhibitor of tyrosine kinases including MET, AXL, and VEGF receptors, was assessed in patients with hepatocellular carcinoma (HCC) as part of a phase 2 randomized discontinuation trial with nine tumor-type cohorts.
Patients and methods: Eligible patients had Child-Pugh A liver function and ≤1 prior systemic anticancer regimen, completed ≥4 weeks before study entry. The cabozantinib starting dose was 100 mg daily. After an initial 12-week cabozantinib treatment period, patients with stable disease (SD) per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.0 were randomized to cabozantinib or placebo. The primary endpoint of the lead-in stage was objective response rate (ORR) at week 12, and the primary endpoint of the randomized stage was progression-free survival (PFS).
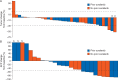
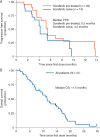
Results: Among the 41 HCC patients enrolled, the week 12 ORR was 5%, with 2 patients achieving a confirmed partial response (PR). The week 12 disease control rate (PR or SD) was 66% (Asian subgroup: 73%). Of patients with ≥1 post-baseline scan, 78% had tumor regression, with no apparent relationship to prior sorafenib therapy. Alpha-fetoprotein (AFP) response (>50% reduction from baseline) occurred in 9 of the 26 (35%) patients with elevated baseline AFP and ≥1 post-baseline measurement. Twenty-two patients with SD at week 12 were randomized. Median PFS after randomization was 2.5 months with cabozantinib and 1.4 months with placebo, although this difference was not statistically significant. Median PFS and overall survival from Day 1 in all patients were 5.2 and 11.5 months, respectively. The most common grade 3/4 adverse events, regardless of attribution, were diarrhea (20%), hand-foot syndrome (15%), and thrombocytopenia (15%). Dose reductions were utilized in 59% of patients.
Conclusions: Cabozantinib has clinical activity in HCC patients, including objective tumor responses, disease stabilization, and reductions in AFP. Adverse events were managed with dose reductions.
Trial registration number: NCT00940225.
Keywords: cabozantinib; hepatocellular carcinoma; overall survival; progression-free survival; tumor response; vascular endothelial growth factor receptor.
© The Author 2017. Published by Oxford University Press on behalf of the European Society for Medical Oncology.
Figures
Comment in
-
Cabozantinib: targeted therapy back to the future?Ann Oncol. 2017 Mar 1;28(3):450-451. doi: 10.1093/annonc/mdx015. Ann Oncol. 2017. PMID: 28137742 No abstract available.
Similar articles
-
Phase II randomised discontinuation trial of cabozantinib in patients with advanced solid tumours.Eur J Cancer. 2017 Nov;86:296-304. doi: 10.1016/j.ejca.2017.09.011. Epub 2017 Oct 20. Eur J Cancer. 2017. PMID: 29059635 Clinical Trial.
-
Comparative Efficacy of Cabozantinib and Regorafenib for Advanced Hepatocellular Carcinoma.Adv Ther. 2020 Jun;37(6):2678-2695. doi: 10.1007/s12325-020-01378-y. Epub 2020 May 18. Adv Ther. 2020. PMID: 32424805 Free PMC article.
-
Results of a Phase II Placebo-controlled Randomized Discontinuation Trial of Cabozantinib in Patients with Non-small-cell Lung Carcinoma.Clin Lung Cancer. 2019 Mar;20(2):74-81.e1. doi: 10.1016/j.cllc.2018.10.006. Epub 2018 Oct 24. Clin Lung Cancer. 2019. PMID: 30528315 Clinical Trial.
-
Sorafenib: a review of its use in advanced hepatocellular carcinoma.Drugs. 2009;69(2):223-40. doi: 10.2165/00003495-200969020-00006. Drugs. 2009. PMID: 19228077 Review.
-
Comparative efficacy and safety for second-line treatment with ramucirumab, regorafenib, and cabozantinib in patients with advanced hepatocellular carcinoma progressed on sorafenib treatment: A network meta-analysis.Medicine (Baltimore). 2021 Sep 24;100(38):e27013. doi: 10.1097/MD.0000000000027013. Medicine (Baltimore). 2021. PMID: 34559096 Free PMC article.
Cited by
-
Hepatocellular Carcinoma: Molecular Pathogenesis and Therapeutic Advances.Cancers (Basel). 2022 Jan 26;14(3):621. doi: 10.3390/cancers14030621. Cancers (Basel). 2022. PMID: 35158892 Free PMC article. Review.
-
Current options and future possibilities for the systemic treatment of hepatocellular carcinoma.Hepat Oncol. 2019 Jun 4;6(1):HEP11. doi: 10.2217/hep-2019-0001. Hepat Oncol. 2019. PMID: 31244990 Free PMC article. Review.
-
Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma.N Engl J Med. 2018 Jul 5;379(1):54-63. doi: 10.1056/NEJMoa1717002. N Engl J Med. 2018. PMID: 29972759 Free PMC article. Clinical Trial.
-
Treatment strategies for hepatocellular carcinoma with extrahepatic metastasis.World J Clin Cases. 2021 Jul 26;9(21):5754-5768. doi: 10.12998/wjcc.v9.i21.5754. World J Clin Cases. 2021. PMID: 34368295 Free PMC article. Review.
-
Systemic Treatment Options in Hepatocellular Carcinoma.Liver Cancer. 2019 Nov;8(6):427-446. doi: 10.1159/000499765. Epub 2019 May 29. Liver Cancer. 2019. PMID: 31799201 Free PMC article. Review.
References
-
- Gherardi E, Birchmeier W, Birchmeier C, Vande Woude G.. Targeting MET in cancer: rationale and progress. Nat Rev Cancer 2012; 12: 89–103. - PubMed
-
- Shojaei F, Lee JH, Simmons BH. et al. HGF/c-Met acts as an alternative angiogenic pathway in sunitinib-resistant tumors. Cancer Res 2010; 70: 10090–10100. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

