CD4 T-cell expression of IFN-γ and IL-17 in pediatric malarial anemia
- PMID: 28426727
- PMCID: PMC5398558
- DOI: 10.1371/journal.pone.0175864
CD4 T-cell expression of IFN-γ and IL-17 in pediatric malarial anemia
Abstract
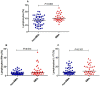
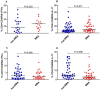
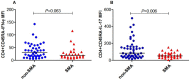
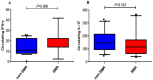
In Plasmodium falciparum holoendemic transmission regions of western Kenya, life-threatening pediatric malaria manifests primarily as severe malarial anemia (SMA, Hb≤6.0 g/dL with any density parasitemia). To determine the role that CD4+ T-cell-driven inflammatory responses have in the pathogenesis of SMA, peripheral CD4+ T-cell populations and their intracellular production of pro-inflammatory cytokines (IFN-γ and IL-17) were characterized in children aged 12-36 months of age stratified into two groups: non-severe malarial anemia (non-SMA, Hb≥6.0 g/dL, n = 50) and SMA (n = 39). In addition, circulating IFN-γ and IL-17 were measured as part of a Cytokine 25-plex Antibody Bead Kit, Human (BioSource™ International). Children with SMA had higher overall proportions of circulating lymphocytes (P = 0.003) and elevated proportions of lymphocytes expressing IFN-γ (P = 0.014) and comparable IL-17 (P = 0.101). In addition, SMA was characterized by decreased memory-like T-cells (CD4+CD45RA-) expressing IL-17 (P = 0.009) and lower mean fluorescence intensity in memory-like CD4+ T-cells for both IFN-γ (P = 0.063) and IL-17 (P = 0.006). Circulating concentrations of IFN-γ were higher in children with SMA (P = 0.009), while IL-17 levels were comparable between the groups (P = 0.164). Furthermore, circulating levels of IFN-γ were negatively correlated with IL-17 levels in both groups of children (SMA: r = -0.610, P = 0.007; and non-SMA: r = -0.516, P = 0.001), while production of both cytokines by lymphocytes were positively correlated (SMA: r = 0.349, P = 0.037; and non-SMA: r = 0.475, P = 0.001). In addition, this correlation was only maintained by the memory-like CD4+ T cells (r = 0.365, P = 0.002) but not the naïve-like CD4+ T cells. However, circulating levels of IFN-γ were only associated with naïve-like CD4+ T cells producing IFN-γ (r = 0.547, P = 0.028), while circulating levels of IL-17 were not associated with any of the cell populations. Taken together, these results suggest that enhanced severity of malarial anemia is associated with higher overall levels of circulating lymphocytes, enhanced intracellular production of IFN-γ by peripheral lymphocytes and high circulating IFN-γ levels. In addition, the observed inverse relationship between the circulating levels of IFN-γ and IL-17 together with the reduction in the levels of memory-like CD4+ T cells expressing IL-17 in children with SMA may suggest possible relocation of these cells in the deeper tissues for their pathological effect.
Conflict of interest statement
Figures

Similar articles
-
Association between Fcγ receptor IIA, IIIA and IIIB genetic polymorphisms and susceptibility to severe malaria anemia in children in western Kenya.BMC Infect Dis. 2017 Apr 20;17(1):289. doi: 10.1186/s12879-017-2390-0. BMC Infect Dis. 2017. PMID: 28427365 Free PMC article.
-
Dynamic changes in peripheral blood CD4+ T cell subsets are associated with litter performance in primiparous gestating sows.J Anim Sci. 2025 Jan 4;103:skaf142. doi: 10.1093/jas/skaf142. J Anim Sci. 2025. PMID: 40293163
-
Differential expression of cytokines and vitamin D in benign and malignant thyroid diseases.Sci Rep. 2025 Jul 2;15(1):23493. doi: 10.1038/s41598-025-06882-1. Sci Rep. 2025. PMID: 40604072 Free PMC article.
-
Effectiveness and safety of vitamin D in relation to bone health.Evid Rep Technol Assess (Full Rep). 2007 Aug;(158):1-235. Evid Rep Technol Assess (Full Rep). 2007. PMID: 18088161 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
Cited by
-
Elevated IL-17 levels in semi-immune anaemic mice infected with Plasmodium berghei ANKA.Malar J. 2018 Apr 17;17(1):169. doi: 10.1186/s12936-018-2257-x. Malar J. 2018. PMID: 29665817 Free PMC article.
-
Autoimmune Anemia in Malaria.Trends Parasitol. 2020 Feb;36(2):91-97. doi: 10.1016/j.pt.2019.12.002. Epub 2019 Dec 18. Trends Parasitol. 2020. PMID: 31864893 Free PMC article. Review.
-
A primate model of severe malarial anaemia: a comparative pathogenesis study.Sci Rep. 2019 Dec 12;9(1):18965. doi: 10.1038/s41598-019-55377-3. Sci Rep. 2019. PMID: 31831787 Free PMC article.
-
Transcriptomic and Proteomic Insights into Host Immune Responses in Pediatric Severe Malarial Anemia: Dysregulation in HSP60-70-TLR2/4 Signaling and Altered Glutamine Metabolism.Pathogens. 2024 Oct 3;13(10):867. doi: 10.3390/pathogens13100867. Pathogens. 2024. PMID: 39452740 Free PMC article.
-
Innate Lymphoid Cells in Protection, Pathology, and Adaptive Immunity During Apicomplexan Infection.Front Immunol. 2019 Feb 28;10:196. doi: 10.3389/fimmu.2019.00196. eCollection 2019. Front Immunol. 2019. PMID: 30873151 Free PMC article. Review.
References
-
- WHO. World Malaria Report 2015. http://wwwwhoint/malaria/publications/world-malaria-report-2015/report/en/. 2015.
-
- WHO. World Malaria Report 2008. [http://wwwwhoint/malaria/wmr2008]. 2008.
-
- Okiro EA, Al-Taiar A, Reyburn H, Idro R, Berkley JA, Snow RW. Age patterns of severe paediatric malaria and their relationship to Plasmodium falciparum transmission intensity. Malar J. 2009;8:4 doi: 10.1186/1475-2875-8-4 - DOI - PMC - PubMed
-
- Roca-Feltrer A, Carneiro I, Smith L, Schellenberg JR, Greenwood B, Schellenberg D. The age patterns of severe malaria syndromes in sub-Saharan Africa across a range of transmission intensities and seasonality settings. Malar J. 2010;9:282 doi: 10.1186/1475-2875-9-282 - DOI - PMC - PubMed
-
- McElroy PD, Lal AA, Hawley WA, Bloland PB, Kuile FO, Oloo AJ, et al. Analysis of repeated hemoglobin measures in full-term, normal birth weight Kenyan children between birth and four years of age. III. The Asembo Bay Cohort Project. Am J Trop Med Hyg. 1999. December;61(6):932–40. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

