IVIg for Treatment of Severe Refractory Heparin-Induced Thrombocytopenia
- PMID: 28427966
- PMCID: PMC5812774
- DOI: 10.1016/j.chest.2017.03.050
IVIg for Treatment of Severe Refractory Heparin-Induced Thrombocytopenia
Abstract
Background: Heparin-induced thrombocytopenia (HIT) complicated by severe thrombocytopenia and thrombosis can pose significant treatment challenges. Use of alternative anticoagulants in this setting may increase bleeding risks, especially in patients who have a protracted disease course. Additional therapies are lacking in this severely affected patient population.
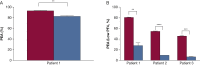
Methods: We describe three patients with HIT who had severe thromboembolism and prolonged thrombocytopenia refractory to standard treatment but who achieved an immediate and sustained response to IVIg therapy. The mechanism of action of IVIg was evaluated in these patients and in five additional patients with severe HIT. The impact of a common polymorphism (H/R 131) in the platelet IgG receptor FcγRIIa on IVIg-mediated inhibition of platelet activation was also examined.
Results: At levels attained in vivo, IVIg inhibits HIT antibody-mediated platelet activation. The constant domain of IgG (Fc) but not the antigen-binding portion (Fab) is required for this effect. Consistent with this finding, IVIg had no effect on HIT antibody binding in a solid-phase HIT immunoassay (platelet factor 4 enzyme-linked immunoassay). The H/R131 polymorphism in FcγRIIa influences the susceptibility of platelets to IVIg treatment, with the HH131 genotype being most susceptible to IVIg-mediated inhibition of antibody-induced activation. However, at high doses of IVIg, activation of platelets of all FcγRIIa genotypes was significantly inhibited. All three patients did well on long-term anticoagulation therapy with direct oral anticoagulants.
Conclusions: These studies suggest that IVIg treatment should be considered in patients with HIT who have severe disease that is refractory to standard therapies.
Keywords: DOAC; HIT; IVIg; heparin; thrombocytopenia; thrombosis.
Copyright © 2017 American College of Chest Physicians. Published by Elsevier Inc. All rights reserved.
Figures



Comment in
-
IV Immunoglobulin for Autoimmune Heparin-Induced Thrombocytopenia.Chest. 2017 Sep;152(3):453-455. doi: 10.1016/j.chest.2017.05.042. Chest. 2017. PMID: 28889872 No abstract available.
-
Intravenous Immunoglobulin for Heparin-Induced Thrombocytopenia.Chest. 2017 Oct;152(4):906-907. doi: 10.1016/j.chest.2017.05.043. Chest. 2017. PMID: 28991551 No abstract available.
References
-
- Greinacher A. Clinical practice. Heparin-induced thrombocytopenia. N Engl J Med. 2015;373(3):252–261. - PubMed
-
- Warkentin T.E., Basciano P.A., Knopman J., Bernstein R.A. Spontaneous heparin-induced thrombocytopenia syndrome: 2 new cases and a proposal for defining this disorder. Blood. 2014;123(23):3651–3654. - PubMed
-
- Rice L., Attisha W.K., Drexler A., Francis J.L. Delayed-onset heparin-induced thrombocytopenia. Ann Intern Med. 2002;136(3):210–215. - PubMed
-
- Warkentin T.E., Kelton J.G. Delayed-onset heparin-induced thrombocytopenia and thrombosis. Ann Intern Med. 2001;135(7):502–506. - PubMed
-
- Warkentin T.E. Agents for the treatment of heparin-induced thrombocytopenia. Hematol Oncol Clin North Am. 2010;24(4):755–775. ix. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

