Deep Brain Stimulation Improves the Symptoms and Sensory Signs of Persistent Central Neuropathic Pain from Spinal Cord Injury: A Case Report
- PMID: 28428749
- PMCID: PMC5382156
- DOI: 10.3389/fnhum.2017.00177
Deep Brain Stimulation Improves the Symptoms and Sensory Signs of Persistent Central Neuropathic Pain from Spinal Cord Injury: A Case Report
Abstract
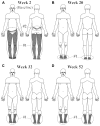
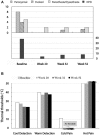
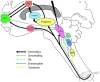
Central neuropathic pain (CNP) is a significant problem after spinal cord injury (SCI). Pharmacological and non-pharmacological approaches may reduce the severity, but relief is rarely substantial. While deep brain stimulation (DBS) has been used to treat various chronic pain types, the technique has rarely been used to attenuate CNP after SCI. Here we present the case of a 54-year-old female with incomplete paraplegia who had severe CNP in the lower limbs and buttock areas since her injury 30 years prior. She was treated with bilateral DBS of the midbrain periaqueductal gray (PAG). The effects of this stimulation on CNP characteristics, severity and pain-related sensory function were evaluated using the International SCI Pain Basic Data Set (ISCIPBDS), Neuropathic Pain Symptom Inventory (NPSI), Multidimensional Pain Inventory and Quantitative Sensory Testing before and periodically after initiation of DBS. After starting DBS treatment, weekly CNP severity ratings rapidly decreased from severe to minimal, paralleled by a substantial reduction in size of the painful area, reduced pain impact and reversal of pain-related neurological abnormalities, i.e., dynamic-mechanical and cold allodynia. She discontinued pain medication on study week 24. The improvement has been consistent. The present study expands on previous findings by providing in-depth assessments of symptoms and signs associated with CNP. The results of this study suggest that activation of endogenous pain inhibitory systems linked to the PAG can eliminate CNP in some people with SCI. More research is needed to better-select appropriate candidates for this type of therapy. We discuss the implications of these findings for understanding the brainstem's control of chronic pain and for future progress in using analgesic DBS in the central gray.
Keywords: chronic pain; evoked pain; low-frequency stimulation; neuromodulation; pain severity; periaqueductal gray.
Figures

Similar articles
-
Spinal Cord Stimulation for Central Neuropathic Pain After Spinal Cord Injury: A Single-Center Case Series.J Pain Res. 2024 Jun 10;17:2029-2035. doi: 10.2147/JPR.S462587. eCollection 2024. J Pain Res. 2024. PMID: 38881761 Free PMC article.
-
Periaqueductal/periventricular gray deep brain stimulation for the treatment of neuropathic facial pain.Front Neurol. 2023 Nov 7;14:1239092. doi: 10.3389/fneur.2023.1239092. eCollection 2023. Front Neurol. 2023. PMID: 38020618 Free PMC article.
-
Corticomotor excitability is altered in central neuropathic pain compared with non-neuropathic pain or pain-free patients.Neurophysiol Clin. 2023 Jun;53(3):102845. doi: 10.1016/j.neucli.2023.102845. Epub 2023 Feb 21. Neurophysiol Clin. 2023. PMID: 36822032
-
Deep brain stimulation of the dorsal anterior cingulate cortex for the treatment of chronic neuropathic pain.Neurosurg Focus. 2015 Jun;38(6):E11. doi: 10.3171/2015.3.FOCUS1543. Neurosurg Focus. 2015. PMID: 26030699 Review.
-
Deep Brain Stimulation and Brain-Spine Interface for Functional Restoration in Spinal Cord Injury.Biomedicines. 2025 Mar 5;13(3):631. doi: 10.3390/biomedicines13030631. Biomedicines. 2025. PMID: 40149607 Free PMC article. Review.
Cited by
-
Cellular Changes in Injured Rat Spinal Cord Following Electrical Brainstem Stimulation.Brain Sci. 2019 May 28;9(6):124. doi: 10.3390/brainsci9060124. Brain Sci. 2019. PMID: 31142050 Free PMC article.
-
Comparative outcomes of microsurgical dorsal root entry zone lesioning (DREZotomy) for intractable neuropathic pain in spinal cord and cauda equina injuries.Neurosurg Rev. 2025 Jan 2;48(1):17. doi: 10.1007/s10143-024-03136-y. Neurosurg Rev. 2025. PMID: 39747752 Free PMC article.
-
A pilot study of neuromuscular electrical stimulation for neuropathic pain caused by spinal cord injury.Medicine (Baltimore). 2018 Aug;97(31):e11658. doi: 10.1097/MD.0000000000011658. Medicine (Baltimore). 2018. PMID: 30075553 Free PMC article.
-
High signal-to-noise imaging of spontaneous and 5 ns electric pulse-evoked Ca2+ signals in GCaMP6f-expressing adrenal chromaffin cells isolated from transgenic mice.PLoS One. 2023 Mar 31;18(3):e0283736. doi: 10.1371/journal.pone.0283736. eCollection 2023. PLoS One. 2023. PMID: 37000822 Free PMC article.
-
Microstructural plasticity in nociceptive pathways after spinal cord injury.J Neurol Neurosurg Psychiatry. 2021 May 26;92(8):863-71. doi: 10.1136/jnnp-2020-325580. Online ahead of print. J Neurol Neurosurg Psychiatry. 2021. PMID: 34039630 Free PMC article.
References
-
- Bobinski F., Ferreira T. A., Córdova M. M., Dombrowski P. A., da Cunha C., Santo C. C., et al. . (2015). Role of brainstem serotonin in analgesia produced by low-intensity exercise on neuropathic pain after sciatic nerve injury in mice. Pain 156, 2595–2606. 10.1097/j.pain.0000000000000372 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

