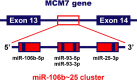
Increased expression of the microRNA 106b~25 cluster and its host gene MCM7 in corticotroph pituitary adenomas is associated with tumor invasion and Crooke's cell morphology
- PMID: 28432562
- PMCID: PMC5508039
- DOI: 10.1007/s11102-017-0805-y
Increased expression of the microRNA 106b~25 cluster and its host gene MCM7 in corticotroph pituitary adenomas is associated with tumor invasion and Crooke's cell morphology
Abstract
Purpose: MCM7 (minichromosome maintenance complex component 7), a DNA replication licensing factor, is a host gene for the oncogenic miR-106b~25 cluster. It has been recently revealed as a relevant prognostic biomarker in a variety of cancers, including pituitary adenomas. The purpose of this study was to assess whether miR-106b~25 and MCM7 levels correlate with tumor invasiveness in a cohort of ACTH-immunopositive adenomas.
Methods: Tissue samples were obtained intraoperatively from 25 patients with pituitary adenoma. Tumor invasiveness was assessed according to the Knosp grading scale. MCM7, Ki-67 and TP53 levels were assessed by immunohistochemical staining, while the expression of miR-106b-5p, miR-93-5p, miR-93-3p and miR-25-3p were measured using quantitative real-time PCR performed on RNA isolated from FFPE tissues.
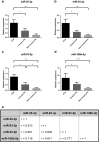
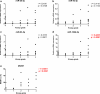
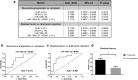
Results: We have found a significant increase in MCM7 and Ki-67 labeling indices in invasive ACTHomas. Moreover, MCM7 was ubiquitously overexpressed in Crooke's cell adenomas. The expression of miR-93-5p was significantly elevated in invasive compared to noninvasive tumors. In addition, all four microRNAs from the miR-106b~25 cluster displayed marked upregulation in Crooke's cell adenomas. Remarkably, MCM7 and miR-106b-5p both strongly correlated with Knosp grade. A combination of MCM7 LI and miR-106b~25 cluster expression was able to accurately differentiate invasive from noninvasive tumors and had a significant discriminatory ability to predict postoperative tumor recurrence/progression.
Conclusions: miR-106b~25 and its host gene MCM7 are potential novel biomarkers for invasive ACTH-immunopositive pituitary adenomas. Additionally, they are both significantly upregulated in rare Crooke's cell adenomas and might therefore contribute to their aggressive phenotype.
Keywords: ACTHoma; Crooke’s cell adenoma; MCM7; MiR-106b~25 cluster; MicroRNA; Pituitary adenoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Minichromosome maintenance protein 7 as prognostic marker of tumor aggressiveness in pituitary adenoma patients.Eur J Endocrinol. 2016 Mar;174(3):307-14. doi: 10.1530/EJE-15-0586. Epub 2015 Nov 30. Eur J Endocrinol. 2016. PMID: 26620390
-
MicroRNA-106b promotes pituitary tumor cell proliferation and invasion through PI3K/AKT signaling pathway by targeting PTEN.Tumour Biol. 2016 Oct;37(10):13469-13477. doi: 10.1007/s13277-016-5155-2. Epub 2016 Jul 27. Tumour Biol. 2016. PMID: 27465551
-
Crooke's cell adenoma of the pituitary: an aggressive variant of corticotroph adenoma.Am J Surg Pathol. 2003 Oct;27(10):1330-6. doi: 10.1097/00000478-200310000-00005. Am J Surg Pathol. 2003. PMID: 14508394
-
Prevalence and clinical characteristics of Crooke's cell adenomas in 101 patients with T-PIT-positive pituitary adenomas: Case series and literature review.Front Endocrinol (Lausanne). 2022 Aug 19;13:947085. doi: 10.3389/fendo.2022.947085. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36060980 Free PMC article. Review.
-
Crooke's cell tumors of the pituitary.Neurosurgery. 2015 May;76(5):616-22. doi: 10.1227/NEU.0000000000000657. Neurosurgery. 2015. PMID: 25635886 Review.
Cited by
-
[Differences in plasma miRNA levels in inferior petrosal sinus samples of patients with ACTH-dependent Cushing's syndrome].Probl Endokrinol (Mosk). 2021 Nov 12;67(6):18-30. doi: 10.14341/probl12817. Probl Endokrinol (Mosk). 2021. PMID: 35018758 Free PMC article. Russian.
-
Identification of Serum microRNA-25 as a novel biomarker for pancreatic cancer.Medicine (Baltimore). 2020 Dec 24;99(52):e23863. doi: 10.1097/MD.0000000000023863. Medicine (Baltimore). 2020. PMID: 33350781 Free PMC article.
-
[Molecular genetic abnormalities in ACTH-secreting pituitary tumors (corticotropinomas): fundamental research and prospects for use in clinical practice].Probl Endokrinol (Mosk). 2023 Nov 2;70(3):23-30. doi: 10.14341/probl13273. Probl Endokrinol (Mosk). 2023. PMID: 39069770 Free PMC article. Review. Russian.
-
Pan-cancer analysis reveals cooperativity of both strands of microRNA that regulate tumorigenesis and patient survival.Nat Commun. 2020 Feb 20;11(1):968. doi: 10.1038/s41467-020-14713-2. Nat Commun. 2020. PMID: 32080184 Free PMC article.
-
circDYRK1A tethers biological behaviors of gastric carcinoma using novel bioinformatics analysis and experimental validations.Sci Rep. 2023 May 22;13(1):8265. doi: 10.1038/s41598-023-33861-1. Sci Rep. 2023. PMID: 37217530 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

