Real-world performance of the new US HIV testing algorithm in medical settings
- PMID: 28434809
- PMCID: PMC5716467
- DOI: 10.1016/j.jcv.2017.04.003
Real-world performance of the new US HIV testing algorithm in medical settings
Abstract
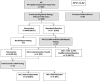
Background: Our medical center laboratory recently adapted its 24/7, two-hourly testing program to use an ARCHITECT-Multispot-viral load (AR-MS-VL) algorithm in place of a previous rapid test-immunofluorescence (RT-IF) algorithm.
Objectives: We evaluated screening test performance, acute case detection, turnaround time and ability to resolve HIV status under the new algorithm.
Study design: We considered consecutive HIV tests from January to November 2015. AR-MS-VL results at Zuckerberg San Francisco General Hospital and Trauma Center (ZSFG) were compared with RT-IF results at ZSFG and also with AR-MS-VL results in the recently completed CDC Screening Targeted Populations to Interrupt On-going Chains of HIV Transmission with Enhanced Partner Notification (STOP) Study for targeted testing of MSM at publicly funded testing sites in San Francisco.
Results: Among 21,985 HIV tests performed at ZSFG, 16,467 were tested by RT-IF and 5518 by AR-MS-VL. There were 321 HIV infections detected, of which 274 (84%) were known HIV+ cases, and 47 were newly identified HIV infections. Considering only patients of HIV-negative or -unknown status, prevalence was 0.22%. Under the AR-MS-VL algorithm, turnaround times for screening results and full algorithm results were 3 and 21h; status-unresolved cases were reduced (from 47% to 22%) compared with the RT-IF algorithm. The positive predictive value (PPV) of a new-positive AR screening test was low (0.44) at ZSFG, where no acute infections were detected. At STOP Study sites where HIV prevalence was higher and acute infection was more common, the AR PPV was higher (0.93). All 24 false-positive AR screening tests at ZSFG had a signal/cutoff (S/CO) ratio of <15 and all 88 true-positive tests had S/CO ratio >15. Of 62 acute infections in the STOP Study, 23 (37%) had an S/CO<15.
Discussion: An AR-MS-VL algorithm is feasible and can return rapid results in a large medical center. In this setting, reactive 4th generation assay tests that are negative for HIV antibodies are typically false-positive with low S/CO ratios.
Keywords: 4th-generation; ARCHITECT; Assay evaluation; Diagnostics; HIV.
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures


References
-
- Branson BM, Handsfield HH, Lampe MA, Janssen RS, Taylor AW, Lyss SB, et al. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR Recomm Rep. 2006;55(RR-14):1–17. - PubMed
-
- Satcher Johnson A, Heitgerd J, Koenig LJ, Van Handel M, Branson BM, Connelly E, et al. Vital signs: HIV testing and diagnosis among adults—United States, 2001–2009. MMWR Morb Mortal Wkly Rep. 2010;59(47):1550–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

