Consequences of the Oculomotor Cycle for the Dynamics of Perception
- PMID: 28434862
- PMCID: PMC5975250
- DOI: 10.1016/j.cub.2017.03.034
Consequences of the Oculomotor Cycle for the Dynamics of Perception
Abstract
Much evidence indicates that humans and other species process large-scale visual information before fine spatial detail. Neurophysiological data obtained with paralyzed eyes suggest that this coarse-to-fine sequence results from spatiotemporal filtering by neurons in the early visual pathway. However, the eyes are normally never stationary: rapid gaze shifts (saccades) incessantly alternate with slow fixational movements. To investigate the consequences of this oculomotor cycle on the dynamics of perception, we combined spectral analysis of visual input signals, neural modeling, and gaze-contingent control of retinal stimulation in humans. We show that the saccade/fixation cycle reformats the flow impinging on the retina in a way that initiates coarse-to-fine processing at each fixation. This finding reveals that the visual system uses oculomotor-induced temporal modulations to sequentially encode different spatial components and suggests that, rather than initiating coarse-to-fine processing, spatiotemporal coupling in the early visual pathway builds on the information dynamics of the oculomotor cycle.
Keywords: coarse-to-fine; contrast sensitivity; fixational eye movements; magnocellular; microsaccade; ocular drift; parvocellular; retina; saccade; spatial vision.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures

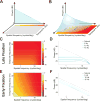



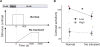
Comment in
-
Active Vision: Dynamic Reformatting of Visual Information by the Saccade-Drift Cycle.Curr Biol. 2017 May 8;27(9):R341-R344. doi: 10.1016/j.cub.2017.03.042. Curr Biol. 2017. PMID: 28486116
References
-
- Robson JG. Spatial and temporal contrast-sensitivity functions of the visual system. J. Opt. Soc. Am. 1966;56:1141–1142.
-
- Nagano T. Temporal sensitivity of the human visual system to sinusoidal gratings. J. Opt. Soc. Am. 1980;70:711–716. - PubMed
-
- Breitmeyer BG, Julesz B. The role of on and off transients in determining the psychophysical spatial frequency response. Vision Res. 1975;15:411–415. - PubMed
-
- Benardete E, Kaplan E. The dynamics of primate M retinal ganglion cells. Visual Neurosci. 1999;16:355–368. - PubMed
-
- Benardete E, Kaplan E. The receptive field of the primate P retinal ganglion cell, I: Linear dynamics. Visual Neurosci. 1997;14:169–185. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

