Randomized Trial of Adding Parenteral Acetaminophen to Prochlorperazine and Diphenhydramine to Treat Headache in the Emergency Department
- PMID: 28435487
- PMCID: PMC5391886
- DOI: 10.5811/westjem.2016.12.29218
Randomized Trial of Adding Parenteral Acetaminophen to Prochlorperazine and Diphenhydramine to Treat Headache in the Emergency Department
Abstract
Introduction: Headaches represent over three million emergency department (ED) visits per year, comprising 2.4% of all ED visits. There are many proposed methods and clinical guidelines of treating acute headache presentations. However, data on intravenous acetaminophen usage in these settings are lacking. In this study, we sought to determine the efficacy of intravenous (IV) acetaminophen as an adjunct to a standard therapy for the treatment of patients who present to the ED with a chief complaint of "headache."
Methods: We conducted a single site, randomized, double-blind, placebo-controlled trial investigating the clinical efficacy of IV acetaminophen as an adjunct to a standard therapy with prochlorperazine and diphenhydramine for the treatment of patients who present to the ED with a chief complaint of "headache" or variants thereof. (See below for variants). The primary outcome measure of the efficacy of parenteral acetaminophen as an adjunct treatment for headache in addition to a standard therapy was a threshold two-point reduction in visual analog scale (VAS) pain scores on a 1-10 level at 90 minutes. Secondary outcomes measures included assessment of decreased requirement of "rescue" pain medicines, defined as any analgesic medications outside of diphenhydramine, prochlorperazine and acetaminophen, with particular interest to potential opioid-sparing effects with parenteral acetaminophen. Additional secondary outcome measure included time to disposition from arrival in the ED.
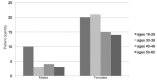
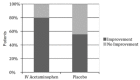
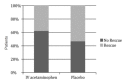
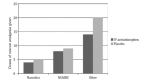
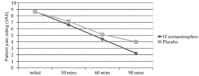
Results: For the acetaminophen group the initial mean pain score was 8.67, for the placebo group 8.61. At 90 minutes pain score was 2.23 for the acetaminophen group and 3.99 for placebo (p<0.01, 95% confidence interval (CI) [0.8%-16%]. Of 45 patients in each group, we observed at least a threshold two-point decrease in pain score 36/45 (80%) with acetaminophen vs. 25/45 (55%) with placebo (p <0.01) 95% CI [5%-41%], number needed to treat (NNT) = 4). Secondary outcome measure did not demonstrate a difference in length of stay (161 minutes for acetaminophen arm and 159 minutes for placebo). However, 17/45 (38%) of patients who received IV acetaminophen required rescue analgesia, opposed to 24/45 (53%) of patients in the placebo group (p=0.13) 95% CI [-5%-34%].
Conclusion: IV acetaminophen when used with prochlorperazine and diphenhydramine to treat acute headaches in the ED resulted in statistically significant pain reduction compared with prochlorperazine and diphenhydramine alone as measured by both threshold of lowering VAS pain score by at least two points (NNT = 4) and overall decline in VAS pain score. Further study is required to validate these results.
Conflict of interest statement
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Figures
Similar articles
-
A prospective, randomized trial of intravenous prochlorperazine versus subcutaneous sumatriptan in acute migraine therapy in the emergency department.Ann Emerg Med. 2010 Jul;56(1):1-6. doi: 10.1016/j.annemergmed.2009.11.020. Epub 2010 Jan 4. Ann Emerg Med. 2010. PMID: 20045576 Clinical Trial.
-
A Comparison of Headache Treatment in the Emergency Department: Prochlorperazine Versus Ketamine.Ann Emerg Med. 2018 Mar;71(3):369-377.e1. doi: 10.1016/j.annemergmed.2017.08.063. Epub 2017 Oct 14. Ann Emerg Med. 2018. PMID: 29033296
-
Diphenhydramine as Adjuvant Therapy for Acute Migraine: An Emergency Department-Based Randomized Clinical Trial.Ann Emerg Med. 2016 Jan;67(1):32-39.e3. doi: 10.1016/j.annemergmed.2015.07.495. Epub 2015 Aug 29. Ann Emerg Med. 2016. PMID: 26320523 Free PMC article. Clinical Trial.
-
The Use of Intravenous Acetaminophen for Acute Pain in the Emergency Department.Acad Emerg Med. 2016 May;23(5):543-53. doi: 10.1111/acem.12921. Epub 2016 Apr 13. Acad Emerg Med. 2016. PMID: 26824905 Review.
-
Intravenous Acetaminophen for Renal Colic in the Emergency Department: Where Do We Stand?Am J Ther. 2017 Jan/Feb;24(1):e12-e19. doi: 10.1097/MJT.0000000000000526. Am J Ther. 2017. PMID: 27779484 Review.
Cited by
-
Acute Pain Management in the Year 2018-A Review.J Acute Med. 2018 Jun 1;8(2):53-59. doi: 10.6705/j.jacme.201806_8(2).0003. J Acute Med. 2018. PMID: 32995204 Free PMC article. Review.
References
-
- Lucado J, Paez K, Elixhauser A. HCUP Statistical Brief #111. Agency for Healthcare Research and Quality; 2011. Headaches in U.S. Hospitals and Emergency Departments, 2008. Available at: http://www.hcup-us.ahrq.gov/reports/statbriefs/sb111.pdf. - PubMed
-
- Taggart E, Doran S, Kokotillo A, et al. Ketorolac in the treatment of acute migraine: a systematic review. Headache. 2013;53(2):277–87. - PubMed
-
- Jones VM. Acetaminophen Injection: A review of clinical information. J Pain Palliat Care Pharmacother. 2012;26(2):115–7. - PubMed
-
- Jahr JS, Lee VK. Intravenous acetaminophen. Anesthesiol Clin. 2010;28(4):619–45. - PubMed
-
- Bektas F, Eken C, Karadeniz O, et al. Intravenous paracetamol or morphine for the treatment of renal colic: A randomized, placebo-controlled trial. Ann Emerg Med. 2009;54(4):568–74. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
