The feline blood film
- PMID: 28438085
- PMCID: PMC10816527
- DOI: 10.1177/1098612X17706466
The feline blood film
Abstract
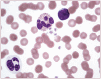
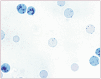
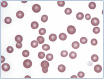
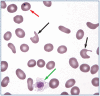
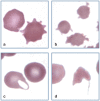
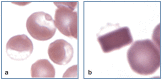
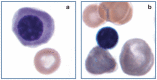
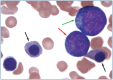
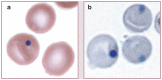
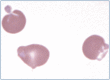
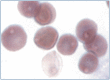
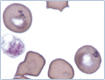
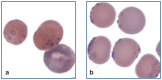
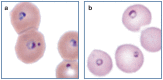
Practical relevance: Many veterinary practices have invested in quality automated hematology instruments for use in-house. However, regardless of the specific choice of analyzer, there are important hematology findings that can only be determined by microscopic examination of stained blood films. For this reason, and also for the purpose of quality control for the analyzer, a quick blood film review should be performed alongside every automated complete blood count. Even those practices that submit their blood samples to outside diagnostic laboratories for evaluation, still require the capability to examine stained blood films in emergency situations. Series outline: This is the first of a two-part article series that aims to familiarize the practitioner with normal findings on feline blood films, with a particular focus on unique features in the cat, as well as to assist with interpretation of common abnormalities. Part 1 focuses on how to prepare and examine blood films in order to maximize the reliability of the information they convey, and describes the morphology of feline erythrocytes in health and disease. Evidence base: The information and guidance offered is based on the published literature and the author's own extensive clinical pathology research.
Conflict of interest statement
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures













References
-
- Harvey JW. Veterinary hematology. A diagnostic guide and color atlas. St Louis, Mo: Elsevier Saunders, 2012.
-
- Mitzner BT. Why automated differentials fall short. J Am Anim Hosp Assoc 2001; 37: 117–118. - PubMed
-
- Bain BJ. Diagnosis from the blood smear. N Engl J Med 2005; 353: 498–507. - PubMed
-
- Welles EG, Hall AS, Carpenter DM. Canine complete blood counts: a comparison of four in-office instruments with the ADVIA 120 and manual differential counts. Vet Clin Pathol 2009; 38: 20–29. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

