Aggregation/dispersion transitions of T4 phage triggered by environmental ion availability
- PMID: 28438164
- PMCID: PMC5404661
- DOI: 10.1186/s12951-017-0266-5
Aggregation/dispersion transitions of T4 phage triggered by environmental ion availability
Abstract
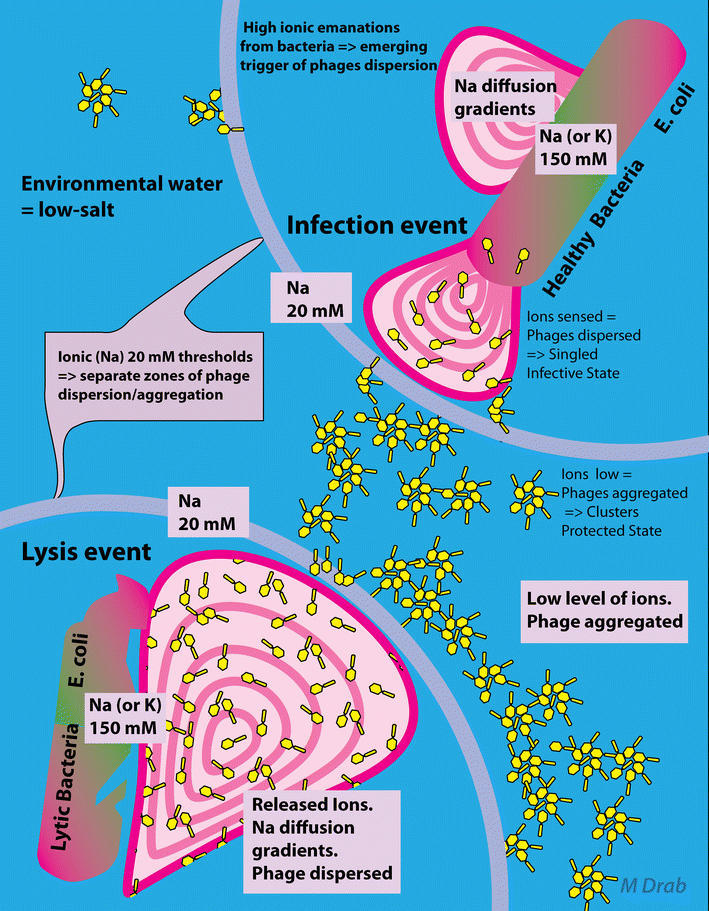
Background: Bacteriophage survives in at least two extremes of ionic environments: bacterial host (high ionic-cytosol) and that of soil (low ionic-environmental water). The impact of ionic composition in the micro- and macro-environments has not so far been addressed in phage biology.
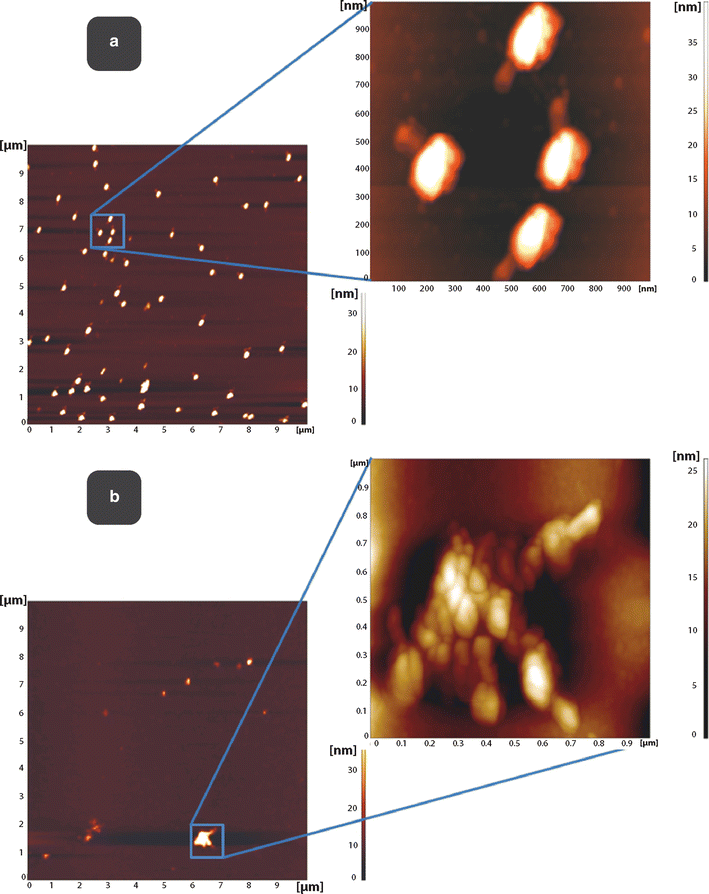
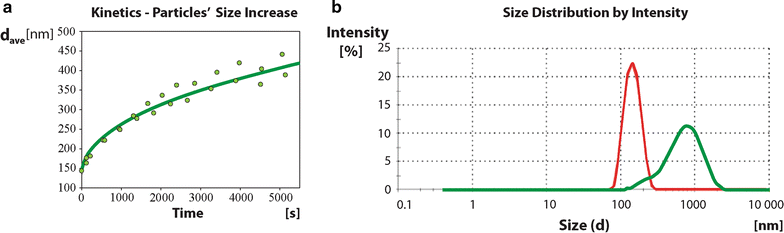
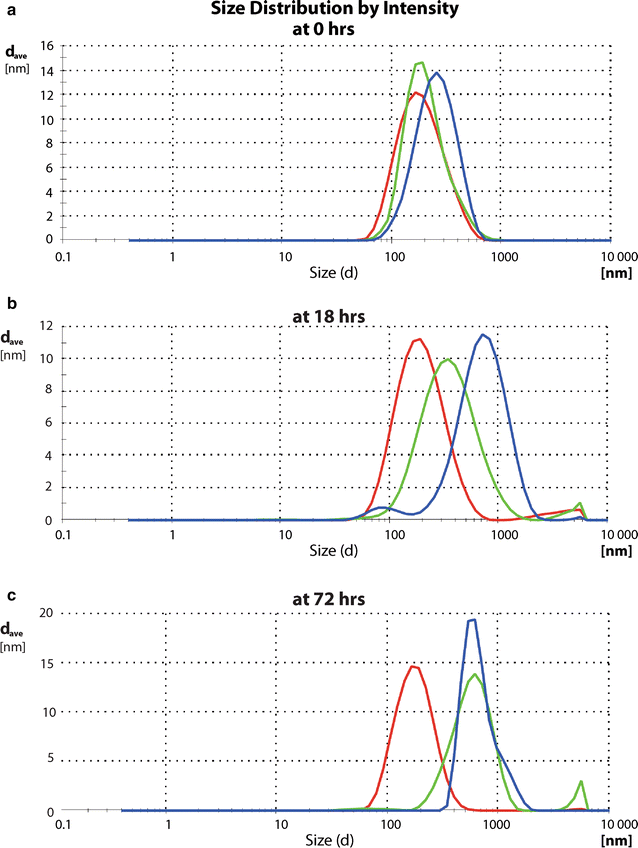
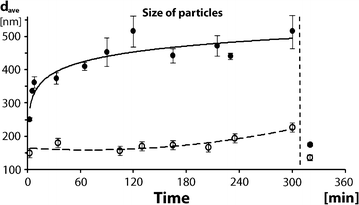
Results: Here, we discovered a novel mechanism of aggregation/disaggregation transitions by phage virions. When normal sodium levels in phage media (150 mM) were lowered to 10 mM, advanced imaging by scanning electron microscopy, atomic force microscopy and dynamic light scattering all revealed formation of viral packages, each containing 20-100 virions. When ionic strength was returned from low to high, the aggregated state of phage reversed to a dispersed state, and the change in ionic strength did not substantially affect infectivity of the phage. By providing the direct evidence, that lowering of the sodium ion below the threshold of 20 mM causes rapid aggregation of phage while returning Na+ concentration to the values above this threshold causes dispersion of phage, we identified a biophysical mechanism of phage aggregation.
Conclusions: Our results implicate operation of group behavior in phage and suggest a new kind of quorum sensing among its virions that is mediated by ions. Loss of ionic strength may act as a trigger in an evolutionary mechanism to improve the survival of bacteriophage by stimulating aggregation of phage when outside a bacterial host. Reversal of phage aggregation is also a promising breakthrough in biotechnological applications, since we demonstrated here the ability to retain viable virion aggregates on standard micro-filters.
Figures

Similar articles
-
Phage Aggregation-Dispersion by Ions: Striving beyond Antibacterial Therapy.Trends Biotechnol. 2018 Sep;36(9):875-881. doi: 10.1016/j.tibtech.2018.03.002. Epub 2018 Apr 10. Trends Biotechnol. 2018. PMID: 29650392 Review.
-
Structural remodeling of bacteriophage T4 and host membranes during infection initiation.Proc Natl Acad Sci U S A. 2015 Sep 1;112(35):E4919-28. doi: 10.1073/pnas.1501064112. Epub 2015 Aug 17. Proc Natl Acad Sci U S A. 2015. PMID: 26283379 Free PMC article.
-
Phage adsorption to bacteria in the light of the electrostatics: a case study using E. coli, T2 and flow cytometry.J Virol Methods. 2013 May;189(2):283-9. doi: 10.1016/j.jviromet.2013.02.007. Epub 2013 Mar 13. J Virol Methods. 2013. PMID: 23499611
-
Effects of environmental and clinical interferents on the host capture efficiency of immobilized bacteriophages.Langmuir. 2014 Mar 25;30(11):3184-90. doi: 10.1021/la500059u. Epub 2014 Mar 11. Langmuir. 2014. PMID: 24617341
-
Bacteriophage-encoded functions engaged in initiation of homologous recombination events.Crit Rev Microbiol. 2009;35(3):197-220. doi: 10.1080/10408410902983129. Crit Rev Microbiol. 2009. PMID: 19563302 Review.
Cited by
-
Real-time monitoring by interferometric light microscopy of phage suspensions for personalised phage therapy.Sci Rep. 2024 Dec 30;14(1):31629. doi: 10.1038/s41598-024-79478-w. Sci Rep. 2024. PMID: 39738265 Free PMC article.
-
Adsorption of bacteriophages on polypropylene labware affects the reproducibility of phage research.Sci Rep. 2021 Apr 1;11(1):7387. doi: 10.1038/s41598-021-86571-x. Sci Rep. 2021. PMID: 33795704 Free PMC article.
-
The Expanding Diversity of Viruses from Extreme Environments.Int J Mol Sci. 2024 Mar 8;25(6):3137. doi: 10.3390/ijms25063137. Int J Mol Sci. 2024. PMID: 38542111 Free PMC article. Review.
-
Extracellular Vesicles and Bacteriophages: New Directions in Environmental Biocolloid Research.Environ Sci Technol. 2023 Nov 7;57(44):16728-16742. doi: 10.1021/acs.est.3c05041. Epub 2023 Oct 29. Environ Sci Technol. 2023. PMID: 37898880 Free PMC article. Review.
-
Nanoencapsulation of Bacteriophages in Liposomes Prepared Using Microfluidic Hydrodynamic Flow Focusing.Front Microbiol. 2018 Sep 12;9:2172. doi: 10.3389/fmicb.2018.02172. eCollection 2018. Front Microbiol. 2018. PMID: 30258426 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

