Structures, Interconversions, and Spectroscopy of Iron Carbonyl Clusters with an Interstitial Carbide: Localized Metal Center Reduction by Overall Cluster Oxidation
- PMID: 28441025
- PMCID: PMC6526005
- DOI: 10.1021/acs.inorgchem.7b00741
Structures, Interconversions, and Spectroscopy of Iron Carbonyl Clusters with an Interstitial Carbide: Localized Metal Center Reduction by Overall Cluster Oxidation
Erratum in
-
Structures, Interconversions, and Spectroscopy of Iron Carbonyl Clusters with an Interstitial Carbide: Localized Metal Center Reduction by Overall Cluster Oxidation.Inorg Chem. 2017 Jul 3;56(13):7575. doi: 10.1021/acs.inorgchem.7b01386. Epub 2017 Jun 12. Inorg Chem. 2017. PMID: 28605580 No abstract available.
Abstract
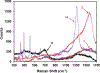
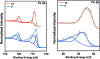
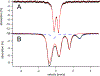
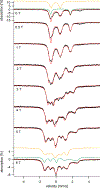
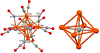
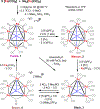
The syntheses, interconversions, and spectroscopic properties of a set of iron carbonyl clusters containing an interstitial carbide are reported. This includes the low temperature X-ray structures of the six-iron clusters (Y)2[Fe6(μ6-C)(μ2-CO)4(CO)12] (1a-c; where Y = NMe4, NEt4, PPh4); the five-iron cluster [Fe5(μ5-C)(CO)15] (3); and the novel formulation of the five-iron cluster (NMe4)2[Fe5(μ5-C)(μ2-CO)(CO)13] (4). Also included in this set is the novel charge-neutral cluster, [Fe6(μ6-C)(CO)18] (2), for which we were unable to obtain a crystallographic structure. As synthetic proof for the identity of 2, we performed a closed loop of interconversions within a family of crystallographically defined species (1, 3, and 4): [Fe6]2- → [Fe6]0 → [Fe5]0 → [Fe5]2- → [Fe6]2-. The structural, spectroscopic, and electronic properties of this "missing link" cluster 2 were investigated by IR, Raman, XPS, and Mössbauer spectroscopies-as well as by DFT calculations. A single νCO feature (1965 cm-1) in the IR spectrum of 2, as well as a prominent Raman feature (νsymm = 1550 cm-1), are consistent with the presence of terminal carbonyls and a {(μ6-C)Fe6} arrangement of iron centers around the central carbide. The XPS of 2 exhibits a higher energy Fe 2p3/2 feature (707.4 eV) as compared to that of 1 (705.5 eV), consistent with the two-electron oxidation induced by treatment of 1 with two equivalents of [Fc](PF6) under CO atmosphere (for the two added CO ligands). DFT calculations indicate two axial and four equatorial Fe sites in 1, all of which have the same or similar oxidation states, for example, two Fe(0) and four Fe(+0.5). These assignments are supported by Mössbauer spectra for 1, which exhibit two closely spaced quadrupole doublets with δ = 0.076 and 0.064 mm s-1. The high-field Mössbauer spectrum of 2 (4.2 K) exhibits three prominent quadrupole doublets with δ = -0.18, -0.11, and +0.41 mm s-1. This indicates three pairs of chemically equivalent Fe sites. The first two pairs arise from irons of a similar oxidation state, while the last pair arises from irons in a different oxidation state, indicating a mixed-valent cluster. Variable field Mössbauer spectra for 2 were simulated assuming these two groups and a diamagnetic ground state. Taken together, the Mössbauer results and DFT calculations for 2 indicate two axial Fe(II) sites and four equatorial sites of lower valence, probably Fe(0). In the DFT optimized pentagonal bipyramidal structure for 2, the Fe(II)-Ccarbide distances are compressed (∼1.84 Å), while the Fe(0)-Ccarbide distances are elongated (∼2.05 Å). Analysis of the formulations for 1 (closo-square bipyramid) and 2 (nido-pentagonal bipyramid) is considered in the context of the textbook electron-counting rules of 14n+2 and 14n+4 for closo and nido clusters, respectively. This redox-dependent intracluster disproportionation of Fe oxidation states is concluded to arise from changes in bonding to the central carbide. A similar phenomenon may be promoted by the central carbide of the FeMoco cluster of nitrogenase, which may in turn stimulate N2 reduction.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
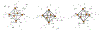
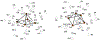
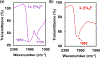
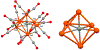

Similar articles
-
Multi-phosphine-chelated iron-carbide clusters via redox-promoted ligand exchange on an inert hexa-iron-carbide carbonyl cluster, [Fe6(μ6-C)(μ2-CO)4(CO)12]2.Chem Sci. 2024 Jun 24;15(29):11455-11471. doi: 10.1039/d4sc01370k. eCollection 2024 Jul 24. Chem Sci. 2024. PMID: 39055015 Free PMC article.
-
Spectroscopic X-ray and Mössbauer Characterization of M6 and M5 Iron(Molybdenum)-Carbonyl Carbide Clusters: High Carbide-Iron Covalency Enhances Local Iron Site Electron Density Despite Cluster Oxidation.Inorg Chem. 2019 Oct 7;58(19):12918-12932. doi: 10.1021/acs.inorgchem.9b01870. Epub 2019 Sep 25. Inorg Chem. 2019. PMID: 31553598 Free PMC article.
-
Single-Step Access to Thiolate-Supported, Six-Iron Clusters with an Authentic Carbide: Synthesis of [Fe6(μ6-C)(μ2-Stol)(μ2-CO)2(CO)12]3 via Non-PSEPT, Redox Intermediates.Inorg Chem. 2025 Aug 25;64(33):16734-16746. doi: 10.1021/acs.inorgchem.4c05362. Epub 2025 Aug 13. Inorg Chem. 2025. PMID: 40802613
-
Mössbauer spectroscopy of Fe/S proteins.Biochim Biophys Acta. 2015 Jun;1853(6):1395-405. doi: 10.1016/j.bbamcr.2014.12.005. Epub 2014 Dec 10. Biochim Biophys Acta. 2015. PMID: 25498248 Review.
-
High-valent iron complexes with tetraamido macrocyclic ligands: structures, Mössbauer spectroscopy, and DFT calculations.J Inorg Biochem. 2006 Apr;100(4):606-19. doi: 10.1016/j.jinorgbio.2005.12.016. Epub 2006 Feb 7. J Inorg Biochem. 2006. PMID: 16464502 Review.
Cited by
-
Quantum chemical studies of redox properties and conformational changes of a four-center iron CO2 reduction electrocatalyst.Chem Sci. 2018 Jan 29;9(10):2645-2654. doi: 10.1039/c7sc04342b. eCollection 2018 Mar 14. Chem Sci. 2018. PMID: 29732050 Free PMC article.
-
From Ylides to Doubly Yldiide-Bridged Iron(II) High Spin Dimers via Self-Protolysis.Inorg Chem. 2019 Jul 15;58(14):9358-9367. doi: 10.1021/acs.inorgchem.9b01086. Epub 2019 Jul 1. Inorg Chem. 2019. PMID: 31260277 Free PMC article.
-
Highly Reduced Ruthenium Carbide Carbonyl Clusters: Synthesis, Molecular Structure, Reactivity, Electrochemistry, and Computational Investigation of [Ru6C(CO)15]4.Inorg Chem. 2023 Sep 11;62(36):14590-14603. doi: 10.1021/acs.inorgchem.3c01711. Epub 2023 Aug 30. Inorg Chem. 2023. PMID: 37646082 Free PMC article.
-
Chemical and Electrochemical Investigation of the Oxidation of a Highly Reduced Fe6C Iron Carbide Carbonyl Cluster: A Synthetic Route to Heteroleptic Fe6C and Fe5C Clusters.Inorg Chem. 2025 May 19;64(19):9744-9757. doi: 10.1021/acs.inorgchem.5c01014. Epub 2025 May 6. Inorg Chem. 2025. PMID: 40327361 Free PMC article.
-
Multi-phosphine-chelated iron-carbide clusters via redox-promoted ligand exchange on an inert hexa-iron-carbide carbonyl cluster, [Fe6(μ6-C)(μ2-CO)4(CO)12]2.Chem Sci. 2024 Jun 24;15(29):11455-11471. doi: 10.1039/d4sc01370k. eCollection 2024 Jul 24. Chem Sci. 2024. PMID: 39055015 Free PMC article.
References
-
- Johnson BFG; Johnston RD; Lewis J Chemistry of polynuclear compounds 14: Hexanuclear carbidocarbonylruthenium compounds. J. Chem. Soc. A 1968, 2865–2868.
-
- Martinengo S; Strumolo D; Chini P Mixed-metal carbide carbonyl clusters 1: Synthesis and structural characterization of di-μ−3-acetonitrile-carbido-ennea-μ-carbonyl-hexacarbonyl-polyhedro-hexarhodium, [Cu2Rh6(CO)15(NCMe)2]0.5MeOH. Inorg. Synth 1980, 20, 212–215.
-
- Hill EW; Bradley JS Transition-metal cluster complexes. Inorg. Synth 1990, 27, 182–188.
-
- Cariati E; Dragonetti C; Lucenti E; Roberto D Tri- and Hexaruthenium carbonyl clusters. Inorg. Synth 2004, 34, 210–214.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

