Collagenous Extracellular Matrix Biomaterials for Tissue Engineering: Lessons from the Common Sea Urchin Tissue
- PMID: 28441344
- PMCID: PMC5454814
- DOI: 10.3390/ijms18050901
Collagenous Extracellular Matrix Biomaterials for Tissue Engineering: Lessons from the Common Sea Urchin Tissue
Abstract
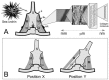
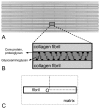
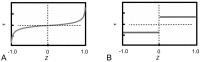
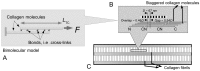
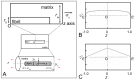
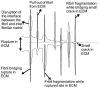
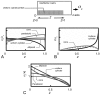
Scaffolds for tissue engineering application may be made from a collagenous extracellular matrix (ECM) of connective tissues because the ECM can mimic the functions of the target tissue. The primary sources of collagenous ECM material are calf skin and bone. However, these sources are associated with the risk of having bovine spongiform encephalopathy or transmissible spongiform encephalopathy. Alternative sources for collagenous ECM materials may be derived from livestock, e.g., pigs, and from marine animals, e.g., sea urchins. Collagenous ECM of the sea urchin possesses structural features and mechanical properties that are similar to those of mammalian ones. However, even more intriguing is that some tissues such as the ligamentous catch apparatus can exhibit mutability, namely rapid reversible changes in the tissue mechanical properties. These tissues are known as mutable collagenous tissues (MCTs). The mutability of these tissues has been the subject of on-going investigations, covering the biochemistry, structural biology and mechanical properties of the collagenous components. Recent studies point to a nerve-control system for regulating the ECM macromolecules that are involved in the sliding action of collagen fibrils in the MCT. This review discusses the key attributes of the structure and function of the ECM of the sea urchin ligaments that are related to the fibril-fibril sliding action-the focus is on the respective components within the hierarchical architecture of the tissue. In this context, structure refers to size, shape and separation distance of the ECM components while function is associated with mechanical properties e.g., strength and stiffness. For simplicity, the components that address the different length scale from the largest to the smallest are as follows: collagen fibres, collagen fibrils, interfibrillar matrix and collagen molecules. Application of recent theories of stress transfer and fracture mechanisms in fibre reinforced composites to a wide variety of collagen reinforcing (non-mutable) connective tissue, has allowed us to draw general conclusions concerning the mechanical response of the MCT at specific mechanical states, namely the stiff and complaint states. The intent of this review is to provide the latest insights, as well as identify technical challenges and opportunities, that may be useful for developing methods for effective mechanical support when adapting decellularised connective tissues from the sea urchin for tissue engineering or for the design of a synthetic analogue.
Keywords: collagen fibril; decellularised tissue; elastic stress transfer; fibril diameter; fibril taper; fibril-fibril interactions; fracture; mutable collagenous tissues; plastic stress transfer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Khang G., Kim M.S., Lee H.B. A Manual for Biomaterials/Scaffold Fabrication Technology. World Scientific; Singapore: 2007.
-
- Williams D. Essential Biomaterials Science. Cambridge University Press; Cambridge, UK: 2014. p. 672.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

