The basis of sharp spike onset in standard biophysical models
- PMID: 28441389
- PMCID: PMC5404793
- DOI: 10.1371/journal.pone.0175362
The basis of sharp spike onset in standard biophysical models
Abstract
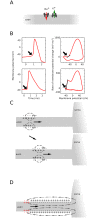
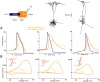
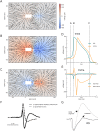
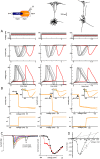
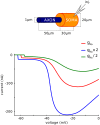
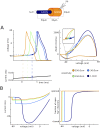
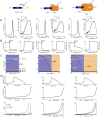
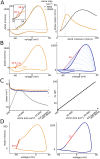
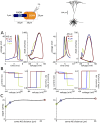
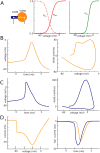
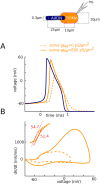
In most vertebrate neurons, spikes initiate in the axonal initial segment (AIS). When recorded in the soma, they have a surprisingly sharp onset, as if sodium (Na) channels opened abruptly. The main view stipulates that spikes initiate in a conventional manner at the distal end of the AIS, then progressively sharpen as they backpropagate to the soma. We examined the biophysical models used to substantiate this view, and we found that spikes do not initiate through a local axonal current loop that propagates along the axon, but through a global current loop encompassing the AIS and soma, which forms an electrical dipole. Therefore, the phenomenon is not adequately modeled as the backpropagation of an electrical wave along the axon, since the wavelength would be as large as the entire system. Instead, in these models, we found that spike initiation rather follows the critical resistive coupling model proposed recently, where the Na current entering the AIS is matched by the axial resistive current flowing to the soma. Besides demonstrating it by examining the balance of currents at spike initiation, we show that the observed increase in spike sharpness along the axon is artifactual and disappears when an appropriate measure of rapidness is used; instead, somatic onset rapidness can be predicted from spike shape at initiation site. Finally, we reproduce the phenomenon in a two-compartment model, showing that it does not rely on propagation. In these models, the sharp onset of somatic spikes is therefore not an artifact of observing spikes at the incorrect location, but rather the signature that spikes are initiated through a global soma-AIS current loop forming an electrical dipole.
Conflict of interest statement
Figures


Similar articles
-
Axonal site of spike initiation enhances auditory coincidence detection.Nature. 2006 Dec 21;444(7122):1069-72. doi: 10.1038/nature05347. Epub 2006 Nov 29. Nature. 2006. PMID: 17136099
-
The determinants of the onset dynamics of action potentials in a computational model.Neuroscience. 2010 Jun 2;167(4):1070-90. doi: 10.1016/j.neuroscience.2010.02.072. Epub 2010 Mar 6. Neuroscience. 2010. PMID: 20211703
-
Electrical match between initial segment and somatodendritic compartment for action potential backpropagation in retinal ganglion cells.J Neurophysiol. 2021 Jul 1;126(1):28-46. doi: 10.1152/jn.00005.2021. Epub 2021 May 26. J Neurophysiol. 2021. PMID: 34038184
-
Neuronal signaling in central nervous system.Sheng Li Xue Bao. 2011 Feb 25;63(1):1-8. Sheng Li Xue Bao. 2011. PMID: 21340428 Review.
-
The electrical significance of axon location diversity.Curr Opin Neurobiol. 2018 Aug;51:52-59. doi: 10.1016/j.conb.2018.02.016. Epub 2018 Mar 10. Curr Opin Neurobiol. 2018. PMID: 29533849 Review.
Cited by
-
Role of sodium channel subtype in action potential generation by neocortical pyramidal neurons.Proc Natl Acad Sci U S A. 2018 Jul 24;115(30):E7184-E7192. doi: 10.1073/pnas.1720493115. Epub 2018 Jul 10. Proc Natl Acad Sci U S A. 2018. PMID: 29991598 Free PMC article.
-
New methods for quantifying rapidity of action potential onset differentiate neuron types.PLoS One. 2021 Apr 8;16(4):e0247242. doi: 10.1371/journal.pone.0247242. eCollection 2021. PLoS One. 2021. PMID: 33831000 Free PMC article.
-
Theory of axo-axonic inhibition.PLoS Comput Biol. 2025 Apr 21;21(4):e1013047. doi: 10.1371/journal.pcbi.1013047. eCollection 2025 Apr. PLoS Comput Biol. 2025. PMID: 40258075 Free PMC article.
-
The location of the axon initial segment affects the bandwidth of spike initiation dynamics.PLoS Comput Biol. 2020 Jul 23;16(7):e1008087. doi: 10.1371/journal.pcbi.1008087. eCollection 2020 Jul. PLoS Comput Biol. 2020. PMID: 32701953 Free PMC article.
-
Tracking axon initial segment plasticity using high-density microelectrode arrays: A computational study.Front Neuroinform. 2022 Oct 3;16:957255. doi: 10.3389/fninf.2022.957255. Front Neuroinform. 2022. PMID: 36221258 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

