Transcriptomic profiling of a chicken lung epithelial cell line (CLEC213) reveals a mitochondrial respiratory chain activity boost during influenza virus infection
- PMID: 28441462
- PMCID: PMC5404788
- DOI: 10.1371/journal.pone.0176355
Transcriptomic profiling of a chicken lung epithelial cell line (CLEC213) reveals a mitochondrial respiratory chain activity boost during influenza virus infection
Abstract
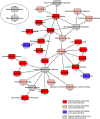
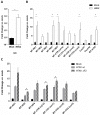
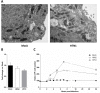
Avian Influenza virus (AIV) is a major concern for the global poultry industry. Since 2012, several countries have reported AIV outbreaks among domestic poultry. These outbreaks had tremendous impact on poultry production and socio-economic repercussion on farmers. In addition, the constant emergence of highly pathogenic AIV also poses a significant risk to human health. In this study, we used a chicken lung epithelial cell line (CLEC213) to gain a better understanding of the molecular consequences of low pathogenic AIV infection in their natural host. Using a transcriptome profiling approach based on microarrays, we identified a cluster of mitochondrial genes highly induced during the infection. Interestingly, most of the regulated genes are encoded by the mitochondrial genome and are involved in the oxidative phosphorylation metabolic pathway. The biological consequences of this transcriptomic induction result in a 2.5- to 4-fold increase of the ATP concentration within the infected cells. PB1-F2, a viral protein that targets the mitochondria was not found associated to the boost of activity of the respiratory chain. We next explored the possibility that ATP may act as a host-derived danger signal (through production of extracellular ATP) or as a boost to increase AIV replication. We observed that, despite the activation of the P2X7 purinergic receptor pathway, a 1mM ATP addition in the cell culture medium had no effect on the virus replication in our epithelial cell model. Finally, we found that oligomycin, a drug that inhibits the oxidative phosphorylation process, drastically reduced the AIV replication in CLEC213 cells, without apparent cellular toxicity. Collectively, our results suggest that AIV is able to boost the metabolic capacities of its avian host in order to provide the important energy needs required to produce progeny virus.
Conflict of interest statement
Figures




Similar articles
-
A novel chicken lung epithelial cell line: characterization and response to low pathogenicity avian influenza virus.Virus Res. 2011 Jul;159(1):32-42. doi: 10.1016/j.virusres.2011.04.022. Epub 2011 May 3. Virus Res. 2011. PMID: 21557972
-
RNA-seq analysis revealed novel genes and signaling pathway associated with disease resistance to avian influenza virus infection in chickens.Poult Sci. 2014 Feb;93(2):485-93. doi: 10.3382/ps.2013-03557. Poult Sci. 2014. PMID: 24570473
-
Susceptibility of primary chicken intestinal epithelial cells for low pathogenic avian influenza virus and velogenic viscerotropic Newcastle disease virus.Virus Res. 2016 Oct 2;225:50-63. doi: 10.1016/j.virusres.2016.09.001. Epub 2016 Sep 3. Virus Res. 2016. PMID: 27596739
-
Avian influenza A viruses in birds --an ecological, ornithological and virological view.Dtsch Tierarztl Wochenschr. 2005 Dec;112(12):448-56. Dtsch Tierarztl Wochenschr. 2005. PMID: 16425630 Review.
-
Poultry food products--a source of avian influenza virus transmission to humans?Clin Microbiol Infect. 2016 Feb;22(2):141-146. doi: 10.1016/j.cmi.2015.11.015. Epub 2015 Dec 12. Clin Microbiol Infect. 2016. PMID: 26686812 Review.
Cited by
-
Transcriptome analysis of chicken intraepithelial lymphocyte natural killer cells infected with very virulent infectious bursal disease virus.Sci Rep. 2020 Oct 27;10(1):18348. doi: 10.1038/s41598-020-75340-x. Sci Rep. 2020. PMID: 33110122 Free PMC article.
-
The Differential Expression of Mitochondrial Function-Associated Proteins and Antioxidant Enzymes during Bovine Herpesvirus 1 Infection: A Potential Mechanism for Virus Infection-Induced Oxidative Mitochondrial Dysfunction.Mediators Inflamm. 2019 Mar 18;2019:7072917. doi: 10.1155/2019/7072917. eCollection 2019. Mediators Inflamm. 2019. PMID: 31011285 Free PMC article.
-
Transcriptomics of chicken cecal tonsils and intestine after infection with low pathogenic avian influenza virus H9N2.Sci Rep. 2021 Oct 14;11(1):20462. doi: 10.1038/s41598-021-99182-3. Sci Rep. 2021. PMID: 34650121 Free PMC article.
-
Influenza A virus directly modulates mouse eosinophil responses.J Leukoc Biol. 2020 Jul;108(1):151-168. doi: 10.1002/JLB.4MA0320-343R. Epub 2020 May 9. J Leukoc Biol. 2020. PMID: 32386457 Free PMC article.
-
Network Meta-Analysis of Chicken Microarray Data following Avian Influenza Challenge-A Comparison of Highly and Lowly Pathogenic Strains.Genes (Basel). 2022 Feb 26;13(3):435. doi: 10.3390/genes13030435. Genes (Basel). 2022. PMID: 35327988 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

