The protein subunit of telomerase displays patterns of dynamic evolution and conservation across different metazoan taxa
- PMID: 28441946
- PMCID: PMC5405514
- DOI: 10.1186/s12862-017-0949-4
The protein subunit of telomerase displays patterns of dynamic evolution and conservation across different metazoan taxa
Abstract
Background: Most animals employ telomerase, which consists of a catalytic subunit known as the telomerase reverse transcriptase (TERT) and an RNA template, to maintain telomere ends. Given the importance of TERT and telomere biology in core metazoan life history traits, like ageing and the control of somatic cell proliferation, we hypothesised that TERT would have patterns of sequence and regulatory evolution reflecting the diverse life histories across the Animal Kingdom.
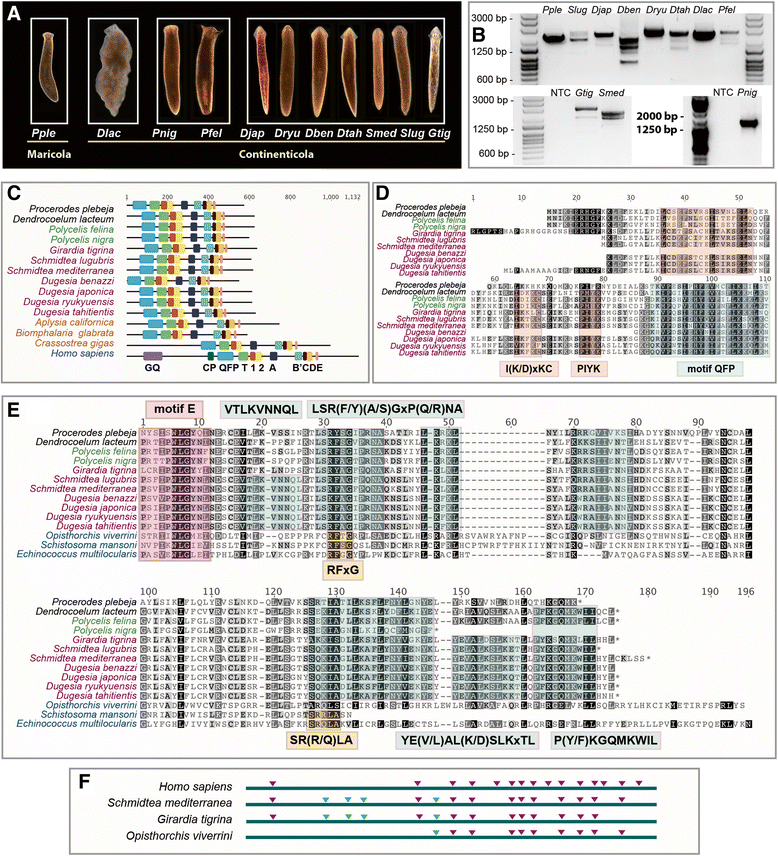
Results: We performed a complete investigation of the evolutionary history of TERT across animals. We show that although TERT is almost ubiquitous across Metazoa, it has undergone substantial sequence evolution within canonical motifs. Beyond the known canonical motifs, we also identify and compare regions that are highly variable between lineages, but show conservation within phyla. Recent data have highlighted the importance of alternative splice forms of TERT in non-canonical functions and although animals may share some conserved introns, we find that the selection of exons for alternative splicing appears to be highly variable, and regulation by alternative splicing appears to be a very dynamic feature of TERT evolution. We show that even within a closely related group of triclad flatworms, where alternative splicing of TERT was previously correlated with reproductive strategy, we observe highly diverse splicing patterns.
Conclusions: Our work establishes that the evolutionary history and structural evolution of TERT involves previously unappreciated levels of change and the emergence of lineage specific motifs. The sequence conservation we describe within phyla suggests that these new motifs likely serve essential biological functions of TERT, which along with changes in splicing, underpin diverse functions of TERT important for animal life histories.
Keywords: Alternative splicing; Evolution; Metazoa; Planarian; TERT; Telomerase.
Figures



Similar articles
-
In silico identification and structural characterization of telomerase reverse transcriptases in parasitic platyhelminths.Gene. 2025 Aug 20;962:149558. doi: 10.1016/j.gene.2025.149558. Epub 2025 May 11. Gene. 2025. PMID: 40360013
-
Structure-function relationships in telomerase genes.Biol Cell. 2009 Jul;101(7):375-92, 1 p following 392. doi: 10.1042/BC20080205. Biol Cell. 2009. PMID: 19419346 Review.
-
The structure and function of telomerase reverse transcriptase.Annu Rev Biochem. 2006;75:493-517. doi: 10.1146/annurev.biochem.75.103004.142412. Annu Rev Biochem. 2006. PMID: 16756500 Review.
-
The role of telomeres and telomerase reverse transcriptase isoforms in pluripotency induction and maintenance.RNA Biol. 2016 Aug 2;13(8):707-19. doi: 10.1080/15476286.2015.1134413. Epub 2016 Jan 19. RNA Biol. 2016. PMID: 26786236 Free PMC article. Review.
-
Structure and function of echinoderm telomerase RNA.RNA. 2016 Feb;22(2):204-15. doi: 10.1261/rna.053280.115. Epub 2015 Nov 23. RNA. 2016. PMID: 26598712 Free PMC article.
Cited by
-
Biogenesis of telomerase RNA from a protein-coding mRNA precursor.Proc Natl Acad Sci U S A. 2022 Oct 11;119(41):e2204636119. doi: 10.1073/pnas.2204636119. Epub 2022 Oct 5. Proc Natl Acad Sci U S A. 2022. PMID: 36197996 Free PMC article.
-
Long-lived termite kings and queens activate telomerase in somatic organs.Proc Biol Sci. 2021 Apr 28;288(1949):20210511. doi: 10.1098/rspb.2021.0511. Epub 2021 Apr 21. Proc Biol Sci. 2021. PMID: 33878922 Free PMC article.
-
A structurally conserved human and Tetrahymena telomerase catalytic core.Proc Natl Acad Sci U S A. 2020 Dec 8;117(49):31078-31087. doi: 10.1073/pnas.2011684117. Epub 2020 Nov 23. Proc Natl Acad Sci U S A. 2020. PMID: 33229538 Free PMC article.
-
A novel flatworm-specific gene implicated in reproduction in Macrostomum lignano.Sci Rep. 2018 Feb 16;8(1):3192. doi: 10.1038/s41598-018-21107-4. Sci Rep. 2018. PMID: 29453392 Free PMC article.
-
Extended longevity of termite kings and queens is accompanied by extranuclear localization of telomerase in somatic organs and caste-specific expression of its isoforms.Insect Sci. 2025 Apr;32(2):364-384. doi: 10.1111/1744-7917.13418. Epub 2024 Jul 21. Insect Sci. 2025. PMID: 39034424 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

