Evaluation of a DNA Aβ42 vaccine in adult rhesus monkeys (Macaca mulatta): antibody kinetics and immune profile after intradermal immunization with full-length DNA Aβ42 trimer
- PMID: 28441965
- PMCID: PMC5405538
- DOI: 10.1186/s13195-017-0257-7
Evaluation of a DNA Aβ42 vaccine in adult rhesus monkeys (Macaca mulatta): antibody kinetics and immune profile after intradermal immunization with full-length DNA Aβ42 trimer
Abstract
Background: Aggregated amyloid-β peptide 1-42 (Aβ42), derived from the cellular amyloid precursor protein, is one of the pathological hallmarks of Alzheimer's disease (AD). Although active immunization against Aβ42 peptide was successful in AD mouse models and led to removal of plaques and improved memory, a similar clinical trial in humans (Aβ42 peptide immunization with QS-21 adjuvant) was stopped in phase II, when 6% of the treated patients developed encephalitis. Currently ongoing passive immunizations with the injection of preformed monoclonal antibodies against different epitopes within the Aβ1-42 peptide, which do not lead to activation of the immune system, have shown some effects in slowing AD pathology. Active DNA Aβ42 immunizations administered with the gene gun into the skin are noninflammatory because they activate a different T-cell population (Th2) with different cytokine responses eliciting a different humoral immune response. We present our findings in rhesus macaques that underwent the DNA Aβ42 immunization via gene gun delivery into the skin.
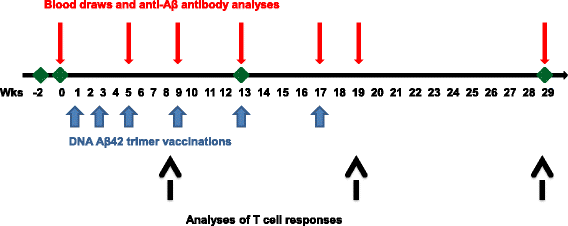
Methods: Six rhesus monkeys received two different doses of a DNA Aβ42 trimer vaccine. The humoral immune response was analyzed from blood throughout the study, and cellular immune responses were determined in peripheral blood mononuclear cells (PBMCs) after three and six immunizations.
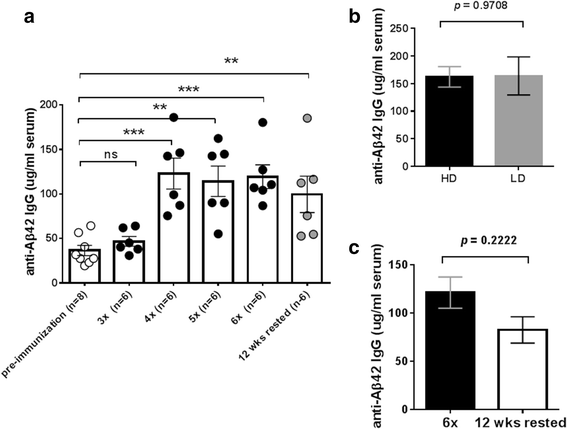
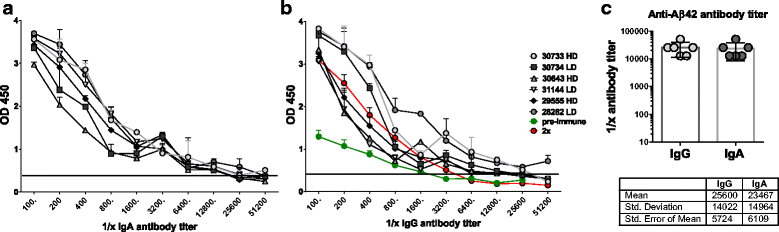
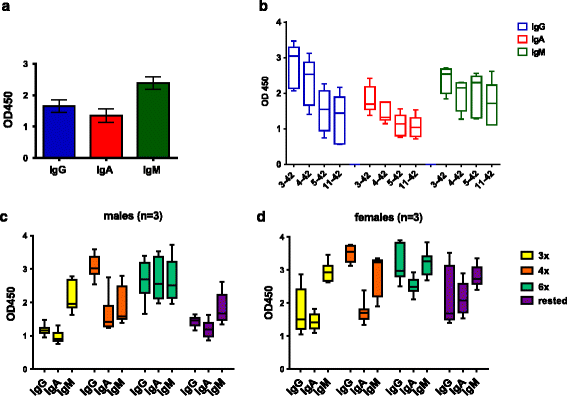
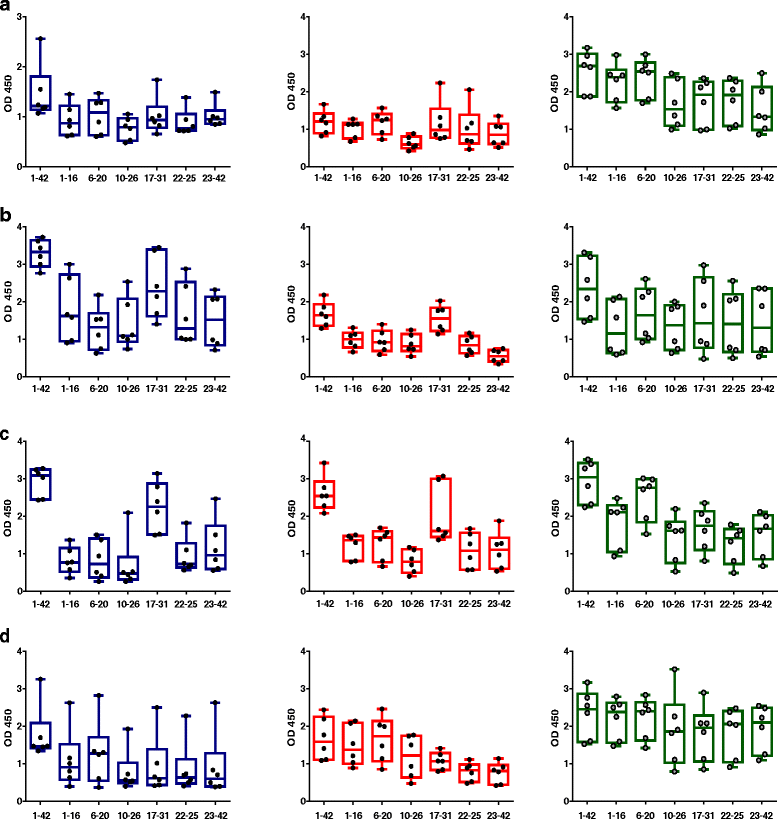
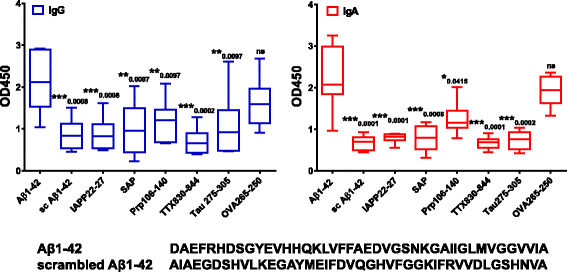
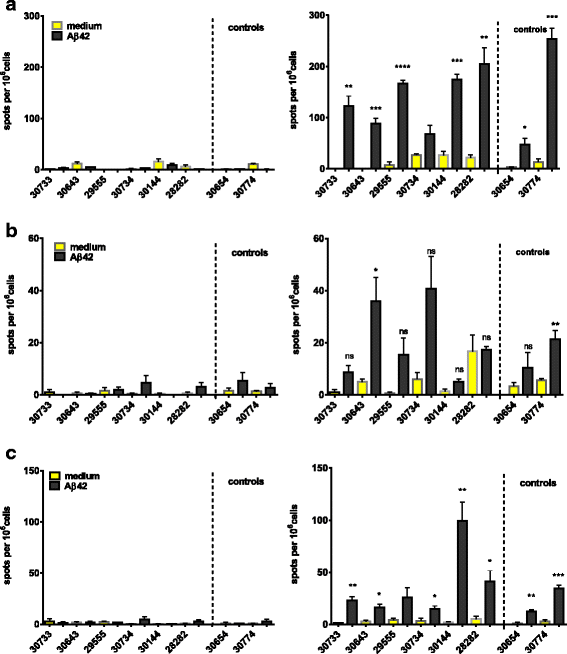
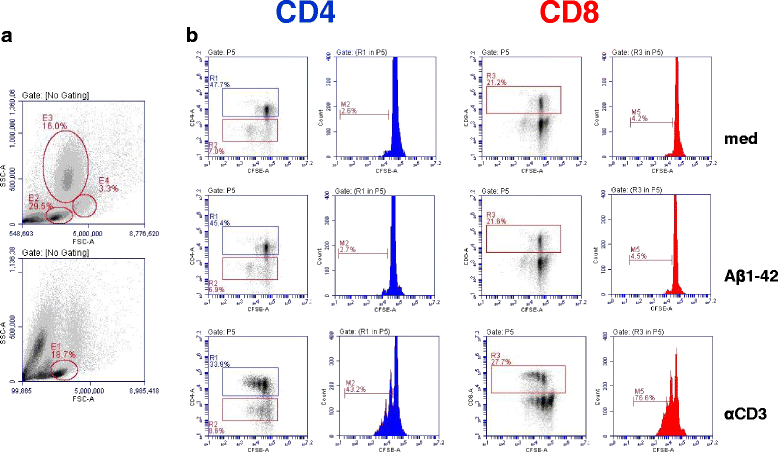
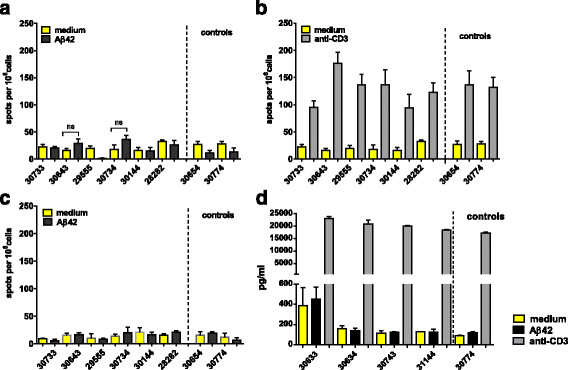
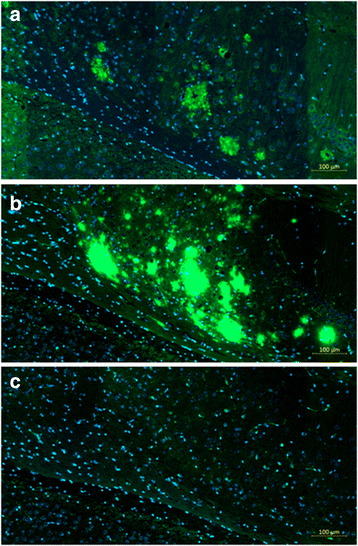
Results: DNA Aβ42 trimer immunization led to high titer antibody responses in the nonhuman primate (NHP) model. Antibodies generated in the rhesus monkeys following DNA Aβ42 immunization detected amyloid plaques consisting of human Aβ42 peptide in the brain of the triple-transgenic AD mouse model. T-cell responses showed no interferon (IFN)-γ- and interleukin (IL)-17-producing cells from PBMCs in Enzyme-Linked ImmunoSpot assays after three immunization time points. At six immunization time points, IFN-γ- and IL-17-producing cells were found in immunized animals as well as in control animals and were thus considered nonspecific and not due to the immunization regimen. IFN-γ and IL-17 secretion in response to Aβ42 peptide restimulation became undetectable after a 3-month rest period.
Conclusions: Intradermal DNA Aβ42 immunization delivered with the gene gun produces a high antibody response in NHPs and is highly likely to be effective and safe in a clinical AD prevention trial in patients.
Keywords: Alzheimer’s disease; Amyloid-β; Antibody response; DNA vaccination; Immunotherapy; Nonhuman primates; Th2 immune response.
Figures
Similar articles
-
Active full-length DNA Aβ42 immunization in 3xTg-AD mice reduces not only amyloid deposition but also tau pathology.Alzheimers Res Ther. 2018 Nov 20;10(1):115. doi: 10.1186/s13195-018-0441-4. Alzheimers Res Ther. 2018. PMID: 30454039 Free PMC article.
-
Evaluation of a DNA Aβ42 Vaccine in Aged NZW Rabbits: Antibody Kinetics and Immune Profile after Intradermal Immunization with Full-Length DNA Aβ42 Trimer.J Alzheimers Dis. 2017;57(1):97-112. doi: 10.3233/JAD-160947. J Alzheimers Dis. 2017. PMID: 28222511 Free PMC article.
-
DNA immunization against amyloid beta 42 has high potential as safe therapy for Alzheimer's disease as it diminishes antigen-specific Th1 and Th17 cell proliferation.Cell Mol Neurobiol. 2011 Aug;31(6):867-74. doi: 10.1007/s10571-011-9680-7. Epub 2011 Apr 6. Cell Mol Neurobiol. 2011. PMID: 21625960 Free PMC article.
-
Developing novel immunogens for a safe and effective Alzheimer's disease vaccine.Prog Brain Res. 2009;175:83-93. doi: 10.1016/S0079-6123(09)17506-4. Prog Brain Res. 2009. PMID: 19660650 Free PMC article. Review.
-
Novel Abeta immunogens: is shorter better?Curr Alzheimer Res. 2007 Sep;4(4):427-36. doi: 10.2174/156720507781788800. Curr Alzheimer Res. 2007. PMID: 17908047 Review.
Cited by
-
Comparative neuropathology in aging primates: A perspective.Am J Primatol. 2021 Nov;83(11):e23299. doi: 10.1002/ajp.23299. Epub 2021 Jul 13. Am J Primatol. 2021. PMID: 34255875 Free PMC article. Review.
-
Active immunotherapy and alternative therapeutic modalities for Alzheimer's disease.Alzheimers Dement (N Y). 2020 Oct 12;6(1):e12090. doi: 10.1002/trc2.12090. eCollection 2020. Alzheimers Dement (N Y). 2020. PMID: 33083513 Free PMC article.
-
Active full-length DNA Aβ42 immunization in 3xTg-AD mice reduces not only amyloid deposition but also tau pathology.Alzheimers Res Ther. 2018 Nov 20;10(1):115. doi: 10.1186/s13195-018-0441-4. Alzheimers Res Ther. 2018. PMID: 30454039 Free PMC article.
-
Nucleic Acid-Based Approaches for Tumor Therapy.Cells. 2020 Sep 9;9(9):2061. doi: 10.3390/cells9092061. Cells. 2020. PMID: 32917034 Free PMC article. Review.
-
Glial cell transcriptome analyses in 3xTg-AD mice: Effects of aging, disease progression, and anti-Aβ immunotherapy.Aging Brain. 2023 Jan 24;3:100066. doi: 10.1016/j.nbas.2023.100066. eCollection 2023. Aging Brain. 2023. PMID: 36911262 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

