Justification of disintegration testing beyond current FDA criteria using in vitro and in silico models
- PMID: 28442890
- PMCID: PMC5395276
- DOI: 10.2147/DDDT.S131213
Justification of disintegration testing beyond current FDA criteria using in vitro and in silico models
Abstract
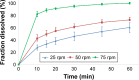
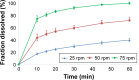
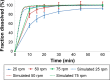
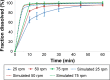
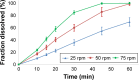
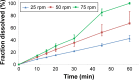
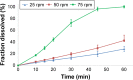
Drug product performance testing is an important part of quality-by-design approaches, but this process often lacks the underlying mechanistic understanding of the complex interactions between the disintegration and dissolution processes involved. Whereas a recent draft guideline by the US Food and Drug Administration (FDA) has allowed the replacement of dissolution testing with disintegration testing, the mentioned criteria are not globally accepted. This study provides scientific justification for using disintegration testing rather than dissolution testing as a quality control method for certain immediate release (IR) formulations. A mechanistic approach, which is beyond the current FDA criteria, is presented. Dissolution testing via United States Pharmacopeial Convention Apparatus II at various paddle speeds was performed for immediate and extended release formulations of metronidazole. Dissolution profile fitting via DDSolver and dissolution profile predictions via DDDPlus™ were performed. The results showed that Fickian diffusion and drug particle properties (DPP) were responsible for the dissolution of the IR tablets, and that formulation factors (eg, coning) impacted dissolution only at lower rotation speeds. Dissolution was completely formulation controlled if extended release tablets were tested and DPP were not important. To demonstrate that disintegration is the most important dosage form attribute when dissolution is DPP controlled, disintegration, intrinsic dissolution and dissolution testing were performed in conventional and disintegration impacting media (DIM). Tablet disintegration was affected by DIM and model fitting to the Korsmeyer-Peppas equation showed a growing effect of the formulation in DIM. DDDPlus was able to predict tablet dissolution and the intrinsic dissolution profiles in conventional media and DIM. The study showed that disintegration has to occur before DPP-dependent dissolution can happen. The study suggests that disintegration can be used as performance test of rapidly disintegrating tablets beyond the FDA criteria. The scientific criteria and justification is that dissolution has to be DPP dependent, originated from active pharmaceutical ingredient characteristics and formulations factors have to be negligible.
Keywords: API; DDDPlus; disintegration; dissolution; model fitting; product specification; quality-by-design.
Conflict of interest statement
Disclosure Gregory K Webster is an employee of AbbVie. The other authors report no conflicts of interest in this work.
Figures
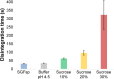


References
-
- Yu LX. Pharmaceutical quality by design: product and process development, understanding, and control. Pharm Res. 2008;25(4):781–791. - PubMed
-
- Almukainzi M, Salehi M, Chacra NAB, Löbenberg R. Comparison of the rupture and disintegration tests for soft-shell capsules. Dissolution Technol. 2011;18(1):21–25.
-
- European Medicines Agency . ICH Topic Q6A: Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances. EMEA; 2000. [Accessed May 6, 2016]. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guidelin.... - PubMed
-
- The United States Pharmacopeia 2016 . USP 39; The national formulary: NF 34. Rockville, MD: United States Pharmacopeial Convention; 2016.
-
- Center for Drug Evaluation and Research (CDER) Guidance for industry: specification criteria for immediate-release solid oral dosage forms containing biopharmaceutics classification system class 1 and 3 Drugs. Food and Drug Administration; 2015. [Accessed May 9, 2016]. Available from: http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformati....
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

