Targeted delivery of thymosin beta 4 to the injured myocardium using CREKA-conjugated nanoparticles
- PMID: 28442910
- PMCID: PMC5396927
- DOI: 10.2147/IJN.S131949
Targeted delivery of thymosin beta 4 to the injured myocardium using CREKA-conjugated nanoparticles
Abstract
Purpose: Thymosin beta 4 (Tβ4) has multiple beneficial facets for myocardial injury, but its efficiency is limited by the low local concentration within the infarct. Here, we established a Tβ4 delivery system for cardiac repair based on the interaction between the abundant fibrin in the infarct zone and the fibrin-targeting moiety clot-binding peptide cysteine-arginine-glutamic acid-lysine-alanine (CREKA).
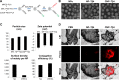
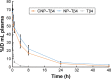
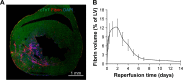
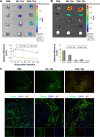
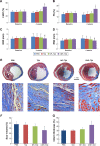
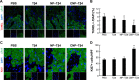
Methods and results: CREKA and Tβ4 were conjugated to nanoparticles (CNP-Tβ4). In vitro binding test revealed that CNP-Tβ4 had a significant binding ability to the surface of fibrin clots when compared to the control clots (NP-Tβ4). Based on the validation of fibrin expression in the early stage of ischemia injury, CNP-Tβ4 was intravenously administered to mice with acute myocardial ischemia-reperfusion injury. CNP-Tβ4 revealed a stronger fibrin-targeting ability than the NP-Tβ4 group and accumulated mainly in the infarcted area and colocalized with fibrin. Subsequently, treatment with CNP-Tβ4 resulted in a better therapeutic effect.
Conclusion: CRKEA modification favored Tβ4 accumulation and retention in the infarcted region, leading to augmented functional benefits. Fibrin-targeting delivery system represents a generalizable platform technology for regenerative medicine.
Keywords: CREKA; cardiovascular; fibrin; ischemia reperfusion; targeting delivery; thymosin beta 4.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures


Similar articles
-
Modification with CREKA Improves Cell Retention in a Rat Model of Myocardial Ischemia Reperfusion.Stem Cells. 2019 May;37(5):663-676. doi: 10.1002/stem.2983. Epub 2019 Mar 12. Stem Cells. 2019. PMID: 30779865
-
Polyethylene glycol-polylactic acid nanoparticles modified with cysteine-arginine-glutamic acid-lysine-alanine fibrin-homing peptide for glioblastoma therapy by enhanced retention effect.Int J Nanomedicine. 2014 Nov 13;9:5261-71. doi: 10.2147/IJN.S72649. eCollection 2014. Int J Nanomedicine. 2014. PMID: 25419130 Free PMC article.
-
Targeted Delivery of Interferon Gamma Using a Recombinant Fusion Protein of a Fibrin Clot-Binding Peptide With Interferon Gamma for Cancer Gene Therapy.J Pharm Sci. 2017 Mar;106(3):892-897. doi: 10.1016/j.xphs.2016.11.018. Epub 2016 Dec 8. J Pharm Sci. 2017. PMID: 27939375
-
Controlled delivery of thymosin β4 for tissue engineering and cardiac regenerative medicine.Ann N Y Acad Sci. 2012 Oct;1269:16-25. doi: 10.1111/j.1749-6632.2012.06718.x. Ann N Y Acad Sci. 2012. PMID: 23045966 Review.
-
Cardioprotection by Thymosin Beta 4.Vitam Horm. 2016;102:209-26. doi: 10.1016/bs.vh.2016.04.004. Epub 2016 May 31. Vitam Horm. 2016. PMID: 27450736 Review.
Cited by
-
Utilizing Developmentally Essential Secreted Peptides Such as Thymosin Beta-4 to Remind the Adult Organs of Their Embryonic State-New Directions in Anti-Aging Regenerative Therapies.Cells. 2021 May 28;10(6):1343. doi: 10.3390/cells10061343. Cells. 2021. PMID: 34071596 Free PMC article. Review.
-
Small non-coding RNA therapeutics for cardiovascular disease.Eur Heart J. 2022 Nov 14;43(43):4548-4561. doi: 10.1093/eurheartj/ehac463. Eur Heart J. 2022. PMID: 36106499 Free PMC article. Review.
-
A literature review of bioactive substances for the treatment of periodontitis: In vitro, in vivo and clinical studies.Heliyon. 2024 Jan 11;10(2):e24216. doi: 10.1016/j.heliyon.2024.e24216. eCollection 2024 Jan 30. Heliyon. 2024. PMID: 38293511 Free PMC article. Review.
-
Biologics and their delivery systems: Trends in myocardial infarction.Adv Drug Deliv Rev. 2021 Jun;173:181-215. doi: 10.1016/j.addr.2021.03.014. Epub 2021 Mar 26. Adv Drug Deliv Rev. 2021. PMID: 33775706 Free PMC article. Review.
-
Advanced Nanomedicine Approaches for Myocardial Infarction Treatment.Int J Nanomedicine. 2024 Jun 24;19:6399-6425. doi: 10.2147/IJN.S467219. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38952676 Free PMC article. Review.
References
-
- Smart N, Risebro CA, Melville AA, et al. Thymosin beta-4 is essential for coronary vessel development and promotes neovascularization via adult epicardium. Ann N Y Acad Sci. 2007;1112:171–188. - PubMed
-
- Smart N, Risebro CA, Melville AA, et al. Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature. 2007;445(7124):177–182. - PubMed
-
- Bock-Marquette I, Saxena A, White MD, Dimaio JM, Srivastava D. Thymosin beta4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair. Nature. 2004;432(7016):466–472. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

