Genetic Deletion of Akt3 Induces an Endophenotype Reminiscent of Psychiatric Manifestations in Mice
- PMID: 28442992
- PMCID: PMC5385361
- DOI: 10.3389/fnmol.2017.00102
Genetic Deletion of Akt3 Induces an Endophenotype Reminiscent of Psychiatric Manifestations in Mice
Abstract
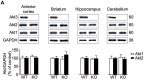
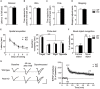
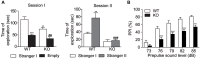
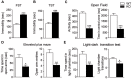
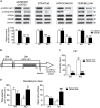
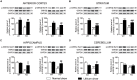
The protein kinase B (PKB/Akt), found in three distinctive isoforms (PKBα/Akt1, PKBβ/Akt2, PKBγ/Akt3), is implicated in a variety of cellular processes such as cell development, growth and survival. Although Akt3 is the most expressed isoform in the brain, its role in cerebral functions is still unclear. In the present study, we investigated the behavioral, electrophysiological and biochemical consequences of Akt3 deletion in mice. Motor abilities, spatial navigation, recognition memory and LTP are intact in the Akt3 knockout (KO) mice. However, the prepulse inhibition, three-chamber social, forced swim, tail suspension, open field, elevated plus maze and light-dark transition tests revealed an endophenotype reminiscent of psychiatric manifestations such as schizophrenia, anxiety and depression. Biochemical investigations revealed that Akt3 deletion was associated with reduced levels of phosphorylated GSK3α/β at serine 21/9 in several brain regions, although Akt1 and Akt2 levels were unaffected. Notably, chronic administration of lithium, a mood stabilizer, restored the decreased phosphorylated GSK3α/β levels and rescued the depressive and anxiety-like behaviors in the Akt3 KO mice. Collectively, our data suggest that Akt3 might be a critical molecule underlying psychiatric-related behaviors in mice.
Keywords: Akt3; GSK3α/β; anxiety; behavior; depression; lithium.
Figures
Similar articles
-
Quantitative analysis of anti-apoptotic function of Akt in Akt1 and Akt2 double knock-out mouse embryonic fibroblast cells under normal and stressed conditions.J Biol Chem. 2006 Oct 20;281(42):31380-8. doi: 10.1074/jbc.M606603200. Epub 2006 Aug 21. J Biol Chem. 2006. PMID: 16923802
-
Akt3 deletion in mice impairs spatial cognition and hippocampal CA1 long long-term potentiation through downregulation of mTOR.Acta Physiol (Oxf). 2019 Jan;225(1):e13167. doi: 10.1111/apha.13167. Epub 2018 Aug 13. Acta Physiol (Oxf). 2019. PMID: 30053339
-
Akt2 and Akt3 play a pivotal role in malignant gliomas.Neuro Oncol. 2010 Mar;12(3):221-32. doi: 10.1093/neuonc/nop026. Epub 2009 Dec 21. Neuro Oncol. 2010. PMID: 20167810 Free PMC article.
-
Promiscuous affairs of PKB/AKT isoforms in metabolism.Arch Physiol Biochem. 2011 May;117(2):70-7. doi: 10.3109/13813455.2010.539236. Epub 2011 Jan 10. Arch Physiol Biochem. 2011. PMID: 21214427 Review.
-
Functional and therapeutic significance of Akt deregulation in malignant melanoma.Cancer Metastasis Rev. 2005 Jun;24(2):273-85. doi: 10.1007/s10555-005-1577-9. Cancer Metastasis Rev. 2005. PMID: 15986137 Review.
Cited by
-
Analysis of Gene Expression Variance in Schizophrenia Using Structural Equation Modeling.Front Mol Neurosci. 2018 Jun 11;11:192. doi: 10.3389/fnmol.2018.00192. eCollection 2018. Front Mol Neurosci. 2018. PMID: 29942251 Free PMC article.
-
Sphingosine-1-Phosphate Receptors Modulators Decrease Signs of Neuroinflammation and Prevent Parkinson's Disease Symptoms in the 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine Mouse Model.Front Pharmacol. 2020 Feb 21;11:77. doi: 10.3389/fphar.2020.00077. eCollection 2020. Front Pharmacol. 2020. PMID: 32153401 Free PMC article.
-
PKBβ/AKT2 deficiency impacts brain mTOR signaling, prefrontal cortical physiology, hippocampal plasticity and select murine behaviors.Mol Psychiatry. 2021 Feb;26(2):411-428. doi: 10.1038/s41380-020-00964-4. Epub 2020 Dec 16. Mol Psychiatry. 2021. PMID: 33328589 Free PMC article.
-
Dual-Specific Protein and Lipid Phosphatase PTEN and Its Biological Functions.Cold Spring Harb Perspect Med. 2020 Jan 2;10(1):a036301. doi: 10.1101/cshperspect.a036301. Cold Spring Harb Perspect Med. 2020. PMID: 31548229 Free PMC article. Review.
-
Akt3 inhibits adipogenesis and protects from diet-induced obesity via WNK1/SGK1 signaling.JCI Insight. 2017 Nov 16;2(22):e95687. doi: 10.1172/jci.insight.95687. eCollection 2017 Nov 16. JCI Insight. 2017. PMID: 29202451 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

