Focal Adhesion Kinase (FAK) tyrosine 397E mutation restores the vascular leakage defect in endothelium-specific FAK-kinase dead mice
- PMID: 28444899
- PMCID: PMC5518444
- DOI: 10.1002/path.4911
Focal Adhesion Kinase (FAK) tyrosine 397E mutation restores the vascular leakage defect in endothelium-specific FAK-kinase dead mice
Abstract
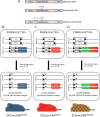
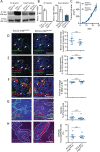
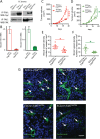
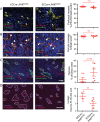
Focal adhesion kinase (FAK) inhibitors have been developed as potential anticancer agents and are undergoing clinical trials. In vitro activation of the FAK kinase domain triggers autophosphorylation of Y397, Src activation, and subsequent phosphorylation of other FAK tyrosine residues. However, how FAK Y397 mutations affect FAK kinase-dead (KD) phenotypes in tumour angiogenesis in vivo is unknown. We developed three Pdgfb-iCreert -driven endothelial cell (EC)-specific, tamoxifen-inducible homozygous mutant mouse lines: FAK wild-type (WT), FAK KD, and FAK double mutant (DM), i.e. KD with a putatively phosphomimetic Y397E mutation. These ECCre+;FAKWT/WT , ECCre+;FAKKD/KD and ECCre+;FAKDM/DM mice were injected subcutaneously with syngeneic B16F0 melanoma cells. Tumour growth and tumour blood vessel functions were unchanged between ECCre+;FAKWT/WT and ECCre-;FAKWT/WT control mice. In contrast, tumour growth and vessel density were decreased in ECCre+;FAKKD/KD and ECCre+;FAKDM/DM mice, as compared with Cre - littermates. Despite no change in the percentage of perfused vessels or pericyte coverage in either genotype, tumour hypoxia was elevated in ECCre+;FAKKD/KD and ECCre+;FAKDM/DM mice. Furthermore, although ECCre+;FAKKD/KD mice showed reduced blood vessel leakage, ECCre+;FAKDM/DM and ECCre-;FAKDM/DM mice showed no difference in leakage. Mechanistically, fibronectin-stimulated Y397 autophosphorylation was reduced in Cre+;FAKKD/KD ECs as compared with Cre+;FAKWT/WT cells, with no change in phosphorylation of the known Src targets FAK-Y577, FAK-Y861, FAK-Y925, paxillin-Y118, p130Cas-Y410. Cre+;FAKDM/DM ECs showed decreased Src target phosphorylation levels, suggesting that the Y397E substitution actually disrupted Src activation. Reduced VE-cadherin-pY658 levels in Cre+;FAKKD/KD ECs were rescued in Cre+FAKDM/DM ECs, corresponding with the rescue in vessel leakage in the ECCre+;FAKDM/DM mice. We show that EC-specific FAK kinase activity is required for tumour growth, angiogenesis, and vascular permeability. The ECCre+;FAKDM/DM mice restored the KD-dependent tumour vascular leakage observed in ECCre+;FAKKD/KD mice in vivo. This study opens new fields in in vivo FAK signalling. © 2017 The Authors. The Journal of Pathology published by John Wiley & Sons Ltd on behalf of Pathological Society of Great Britain and Ireland.
Keywords: focal adhesion kinase; tumour angiogenesis.
© 2017 The Authors. The Journal of Pathology published by John Wiley & Sons Ltd on behalf of Pathological Society of Great Britain and Ireland.
Figures


References
-
- Lechertier T, Hodivala‐Dilke K. Focal adhesion kinase and tumour angiogenesis. J Pathol 2012; 226 : 404–412. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

