Prostaglandin E2 produced following infection with Theiler's virus promotes the pathogenesis of demyelinating disease
- PMID: 28445497
- PMCID: PMC5406002
- DOI: 10.1371/journal.pone.0176406
Prostaglandin E2 produced following infection with Theiler's virus promotes the pathogenesis of demyelinating disease
Abstract
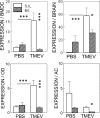
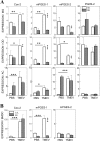
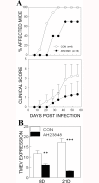
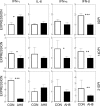
Infection of various cells with Theiler's murine encephalomyelitis virus (TMEV) activates the TLR- and melanoma differentiation-associated gene 5 (MDA5)-dependent pathways, resulting in the production of IL-1β via the activation of caspase-1 upon assembly of the node-like receptor protein 3 (NLRP3) inflammasome. The role of IL-1β in the pathogenesis of TMEV-induced demyelinating disease was previously investigated. However, the signaling effects of prostaglandin E2 (PGE2) downstream of the NLRP3 inflammasome on the immune responses to viral determinants and the pathogenesis of demyelinating disease are unknown. In this study, we investigated the levels of intermediate molecules leading to PGE2 signaling and the effects of blocking PGE2 signaling on the immune response to TMEV infection, viral persistence and the development of demyelinating disease. We demonstrate here that TMEV infection activates the NLRP3 inflammasome and PGE2 signaling much more vigorously in dendritic cells (DCs) and CD11b+ cells from susceptible SJL mice than in cells from resistant B6 mice. Inhibition of virus-induced PGE2 signaling using AH23848 resulted in decreased pathogenesis of demyelinating disease and viral loads in the central nervous system (CNS). In addition, AH23848 treatment caused the elevation of protective early IFN-γ-producing CD4+ and CD8+ T cell responses. Because the levels of IFN-β were lower in AH23848-treated mice but the level of IL-6 was similar, over-production of pathogenic IFN-β was modulated and the generation of IFN-γ-producing T cell responses was enhanced by the inhibition of PGE2 signaling. These results strongly suggest that excessive activation of the NLRP3 inflammasome and downstream PGE2 signaling contribute to the pathogenesis of TMEV-induced demyelinating disease.
Conflict of interest statement
Figures



Similar articles
-
Excessive Innate Immunity Steers Pathogenic Adaptive Immunity in the Development of Theiler's Virus-Induced Demyelinating Disease.Int J Mol Sci. 2021 May 17;22(10):5254. doi: 10.3390/ijms22105254. Int J Mol Sci. 2021. PMID: 34067536 Free PMC article. Review.
-
Critical role of TLR activation in viral replication, persistence, and pathogenicity of Theiler's virus.Front Immunol. 2023 Apr 20;14:1167972. doi: 10.3389/fimmu.2023.1167972. eCollection 2023. Front Immunol. 2023. PMID: 37153539 Free PMC article. Review.
-
TLR3 signaling is either protective or pathogenic for the development of Theiler's virus-induced demyelinating disease depending on the time of viral infection.J Neuroinflammation. 2011 Dec 21;8:178. doi: 10.1186/1742-2094-8-178. J Neuroinflammation. 2011. PMID: 22189096 Free PMC article.
-
The level of viral infection of antigen-presenting cells correlates with the level of development of Theiler's murine encephalomyelitis virus-induced demyelinating disease.J Virol. 2015 Feb;89(3):1867-78. doi: 10.1128/JVI.02471-14. Epub 2014 Nov 26. J Virol. 2015. PMID: 25428872 Free PMC article.
-
Effects of Keratinocyte-Derived Cytokine (CXCL-1) on the Development of Theiler's Virus-Induced Demyelinating Disease.Front Cell Infect Microbiol. 2018 Jan 23;8:9. doi: 10.3389/fcimb.2018.00009. eCollection 2018. Front Cell Infect Microbiol. 2018. PMID: 29410948 Free PMC article.
Cited by
-
Excessive Innate Immunity Steers Pathogenic Adaptive Immunity in the Development of Theiler's Virus-Induced Demyelinating Disease.Int J Mol Sci. 2021 May 17;22(10):5254. doi: 10.3390/ijms22105254. Int J Mol Sci. 2021. PMID: 34067536 Free PMC article. Review.
-
IFN-β Deficiency Results in Fatal or Demyelinating Disease in C57BL/6 Mice Infected With Theiler's Murine Encephalomyelitis Viruses.Front Immunol. 2022 Feb 9;13:786940. doi: 10.3389/fimmu.2022.786940. eCollection 2022. Front Immunol. 2022. PMID: 35222374 Free PMC article.
-
Critical role of TLR activation in viral replication, persistence, and pathogenicity of Theiler's virus.Front Immunol. 2023 Apr 20;14:1167972. doi: 10.3389/fimmu.2023.1167972. eCollection 2023. Front Immunol. 2023. PMID: 37153539 Free PMC article. Review.
-
Infection and Activation of B Cells by Theiler's Murine Encephalomyelitis Virus (TMEV) Leads to Autoantibody Production in an Infectious Model of Multiple Sclerosis.Cells. 2020 Jul 27;9(8):1787. doi: 10.3390/cells9081787. Cells. 2020. PMID: 32727036 Free PMC article.
-
ASC- and caspase-1-deficient C57BL/6 mice do not develop demyelinating disease after infection with Theiler's murine encephalomyelitis virus.Sci Rep. 2023 Jul 6;13(1):10960. doi: 10.1038/s41598-023-38152-3. Sci Rep. 2023. PMID: 37414913 Free PMC article.
References
-
- So EY, Kang MH, Kim BS. Induction of chemokine and cytokine genes in astrocytes following infection with Theiler's murine encephalomyelitis virus is mediated by the Toll-like receptor 3. Glia. 2006;53(8):858–67. doi: 10.1002/glia.20346 - DOI - PubMed
-
- So EY, Kim BS. Theiler's virus infection induces TLR3-dependent upregulation of TLR2 critical for proinflammatory cytokine production. Glia. 2009;57:1216–26. Epub 2009/02/05. doi: 10.1002/glia.20843 - DOI - PMC - PubMed
-
- Jin YH, Kim SJ, So EY, Meng L, Colonna M, Kim BS. Melanoma differentiation-associated gene 5 is critical for protection against Theiler's virus-induced demyelinating disease. J Virol. 2012;86(3):1531–43. Epub 2011/11/18. PubMed Central PMCID: PMC3264388. doi: 10.1128/JVI.06457-11 - DOI - PMC - PubMed
-
- Kanneganti TD, Body-Malapel M, Amer A, Park JH, Whitfield J, Franchi L, et al. Critical role for Cryopyrin/Nalp3 in activation of caspase-1 in response to viral infection and double-stranded RNA. J Biol Chem. 2006;281(48):36560–8. doi: 10.1074/jbc.M607594200 - DOI - PubMed
-
- Ichinohe T, Lee HK, Ogura Y, Flavell R, Iwasaki A. Inflammasome recognition of influenza virus is essential for adaptive immune responses. J Exp Med. 2009;206(1):79–87. PubMed Central PMCID: PMCPMC2626661. doi: 10.1084/jem.20081667 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

