Therapeutic efficacy of artemether-lumefantrine against uncomplicated Plasmodium falciparum malaria in a high-transmission area in northwest Ethiopia
- PMID: 28445503
- PMCID: PMC5405980
- DOI: 10.1371/journal.pone.0176004
Therapeutic efficacy of artemether-lumefantrine against uncomplicated Plasmodium falciparum malaria in a high-transmission area in northwest Ethiopia
Abstract
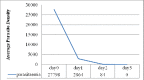
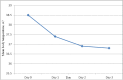
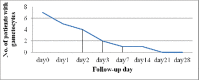
Malaria, particularly due to Plasmodium falciparum, remains a major public health threat in Ethiopia. Artemether-lumefantine (AL) has been the first-line antimalarial drug against uncomplicated P. falciparum malaria in the country since 2004. Regular monitoring of antimalarial drugs is recommended by the World Health Organization (WHO) to help early detection of drug resistant strains of the parasite and contain their rapid spread. The objective of this study was to assess the therapeutic efficacy of AL in a high-transmission setting in Ethiopia. The study site was Setit Humera, northwest Ethiopia. Single-arm prospective study of a 28-day follow-up was conducted from October 2014 to January 2015 according to the revised WHO 2009 drug efficacy study protocol. Study end-points were classified into primary end-point and secondary end-point. While the primary end-point was the day-28 adequate clinical and parasitological response the secondary end-points were clinical and parasitological evaluations (parasite, fever and gametocyte clearance rate, incidence of drug adverse events) and the relative increment in hemoglobin (Hb) level from baseline to day (D) 14 and D28. A total of 92 patients were enrolled and 79 had completed the 28-day follow-up period. The overall cure rate was 98.8% with 95% confidence interval of 0.915-0.998 without polymerase chain reaction correction. The parasite clearance rate was high with fast resolution of clinical symptoms; 100% of the study participants cleared parasitaemia and fever on D3. Gametocyte carriage was reduced from 7% on D0 to 1% on D3 and complete clearance was achieved on D14. Mean Hb concentration significantly increased on D28 compared to that on D14. There was no serious adverse event. AL was efficacious and safe in a high-transmission setting for treatment of uncomplicated falciparum malaria.
Conflict of interest statement
Figures
Similar articles
-
Therapeutic efficacy and safety of artemether-lumefantrine for uncomplicated Plasmodium falciparum malaria treatment in Metehara, Central-east Ethiopia.Malar J. 2024 Jun 12;23(1):184. doi: 10.1186/s12936-024-04991-2. Malar J. 2024. PMID: 38867217 Free PMC article.
-
Efficacy of artemether-lumefantrine therapy for the treatment of uncomplicated Plasmodium falciparum malaria in Southwestern Ethiopia.Malar J. 2015 Aug 15;14:317. doi: 10.1186/s12936-015-0826-9. Malar J. 2015. PMID: 26271736 Free PMC article. Clinical Trial.
-
Therapeutic efficacy of artemether-lumefantrine on treatment of uncomplicated Plasmodium falciparum mono-infection in an area of high malaria transmission in Zambia.Malar J. 2014 Nov 17;13:430. doi: 10.1186/1475-2875-13-430. Malar J. 2014. PMID: 25403945 Free PMC article.
-
Therapeutic efficacy of artemether-lumefantrine in the treatment of uncomplicated Plasmodium falciparum malaria in Ethiopia: a systematic review and meta-analysis.Infect Dis Poverty. 2017 Nov 15;6(1):157. doi: 10.1186/s40249-017-0372-5. Infect Dis Poverty. 2017. PMID: 29137664 Free PMC article.
-
Artemether-lumefantrine: an option for malaria.Ann Pharmacother. 2012 Apr;46(4):567-77. doi: 10.1345/aph.1Q539. Epub 2012 Apr 10. Ann Pharmacother. 2012. PMID: 22496476 Review.
Cited by
-
Persistence of Residual Submicroscopic P. falciparum Parasitemia following Treatment of Artemether-Lumefantrine in Ethio-Sudan Border, Western Ethiopia.Antimicrob Agents Chemother. 2022 Sep 20;66(9):e0000222. doi: 10.1128/aac.00002-22. Epub 2022 Aug 22. Antimicrob Agents Chemother. 2022. PMID: 35993723 Free PMC article.
-
Increasing day three parasitaemia is observed after treatment of patients with artemether-lumefantrine and single dose of primaquine for uncomplicated Plasmodium falciparum malaria in Arbaminch Zuria district, Southwest Ethiopia.Malar J. 2025 Mar 18;24(1):89. doi: 10.1186/s12936-025-05337-2. Malar J. 2025. PMID: 40102850 Free PMC article.
-
Comparison of malaria treatment outcome of generic and innovator's anti-malarial drugs containing artemether-lumefantrine combination in the management of uncomplicated malaria amongst Tanzanian children.Malar J. 2019 Apr 11;18(1):133. doi: 10.1186/s12936-019-2769-z. Malar J. 2019. PMID: 30975147 Free PMC article. Clinical Trial.
-
Comparing the diagnostic accuracy of the newly introduced and existing malaria tests in Northwest Ethiopia.PLoS One. 2025 May 7;20(5):e0322366. doi: 10.1371/journal.pone.0322366. eCollection 2025. PLoS One. 2025. PMID: 40333915 Free PMC article.
-
Therapeutic efficacy and safety of artemether-lumefantrine for uncomplicated Plasmodium falciparum malaria treatment in Metehara, Central-east Ethiopia.Malar J. 2024 Jun 12;23(1):184. doi: 10.1186/s12936-024-04991-2. Malar J. 2024. PMID: 38867217 Free PMC article.
References
-
- WHO: World malaria report 2015 World Health Organization; Geneva, Switzerland, 2015.
-
- Darymple U, Mappin B, Gething PW: Malaria mapping: understanding the global endemicity of falciparum and vivax malaria. BMC Medicine 2015, 13:140 doi: 10.1186/s12916-015-0372-x - DOI - PMC - PubMed
-
- Hemingway J, Shretta R, Wells CNT, Bell D, Djimdé AA, Achee N et al.: Tools and strategies for malaria control and elimination: What do we need to achieve a grand convergence in malaria? PLoS Biol 2016, 14(3):e1002380 doi: 10.1371/journal.pbio.1002380 - DOI - PMC - PubMed
-
- WHO global insecticide resistance database. [http://www.who.int/malaria/areas/vector_control/insecticide_resistance_d...]. Accessed 25 October 2016.
-
- Fillinger U, Lindsay S: Larval source management for malaria control in Africa; myths and reality. Malar J 2011, 10:353 doi: 10.1186/1475-2875-10-353 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources