Transplantation of induced mesenchymal stem cells for treating chronic renal insufficiency
- PMID: 28445516
- PMCID: PMC5405955
- DOI: 10.1371/journal.pone.0176273
Transplantation of induced mesenchymal stem cells for treating chronic renal insufficiency
Abstract
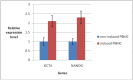
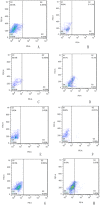
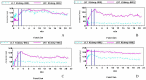
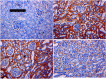
Discovering a new cell transplantation approach for treating chronic renal insufficiency is a goal of many nephrologists. In vitro-cultured peripheral blood mononuclear cells (PBMCs) were reprogrammed into induced mesenchymal stem cells (iMSCs) by using natural inducing agents made in our laboratory. The stem cell phenotype of the iMSCs was then identified. Unilateral ureteral obstruction (UUO) was used to create an animal model of chronic renal insufficiency characterized by renal interstitial fibrosis. The induced and non-induced PBMCs were transplanted, and the efficacy of iMSCs in treating chronic renal insufficiency was evaluated using a variety of methods. The ultimate goal was to explore the effects of iMSC transplantation on the treatment of chronic renal insufficiency, with the aim of providing a new therapeutic modality for this disease.
Conflict of interest statement
Figures


Similar articles
-
Exosomes derived from mesenchymal stem cells ameliorate renal fibrosis via delivery of miR-186-5p.Hum Cell. 2022 Jan;35(1):83-97. doi: 10.1007/s13577-021-00617-w. Epub 2021 Sep 28. Hum Cell. 2022. PMID: 34585365
-
Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells from the Tasmanian Devil (Sarcophilus harrisii) Express Immunomodulatory Factors and a Tropism Toward Devil Facial Tumor Cells.Stem Cells Dev. 2020 Jan 1;29(1):25-37. doi: 10.1089/scd.2019.0203. Stem Cells Dev. 2020. PMID: 31709909
-
Human umbilical cord-derived mesenchymal stem cells conditioned medium attenuate interstitial fibrosis and stimulate the repair of tubular epithelial cells in an irreversible model of unilateral ureteral obstruction.Nephrology (Carlton). 2018 Aug;23(8):728-736. doi: 10.1111/nep.13099. Nephrology (Carlton). 2018. PMID: 28667820
-
Bone marrow-derived mesenchymal stem cells attenuate tubulointerstitial injury through multiple mechanisms in UUO model.J Cell Biochem. 2019 Jun;120(6):9737-9746. doi: 10.1002/jcb.28254. Epub 2018 Dec 7. J Cell Biochem. 2019. PMID: 30525227
-
An approach to treating older adults with chronic kidney disease.CMAJ. 2023 May 1;195(17):E612-E618. doi: 10.1503/cmaj.221427. CMAJ. 2023. PMID: 37127307 Free PMC article. Review. No abstract available.
Cited by
-
Mesenchymal Stem Cells in Renal Fibrosis: The Flame of Cytotherapy.Stem Cells Int. 2019 Jan 13;2019:8387350. doi: 10.1155/2019/8387350. eCollection 2019. Stem Cells Int. 2019. PMID: 30766607 Free PMC article. Review.
-
Recent Updates on Mesenchymal Stem Cell Based Therapy for Acute Renal Failure.Curr Urol. 2020 Jan;13(4):189-199. doi: 10.1159/000499272. Epub 2020 Jan 7. Curr Urol. 2020. PMID: 31998051 Free PMC article. Review.
-
Advancements and Innovative Strategies in Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cell Therapy: A Comprehensive Review.Stem Cells Int. 2024 Sep 30;2024:4073485. doi: 10.1155/2024/4073485. eCollection 2024. Stem Cells Int. 2024. PMID: 39377039 Free PMC article. Review.
-
Evaluation of the efficacy of FDG-PET/CT in assessing the effects of BMMSC treatment in aged rhesus monkeys.Stem Cell Res Ther. 2025 Aug 22;16(1):448. doi: 10.1186/s13287-025-04533-w. Stem Cell Res Ther. 2025. PMID: 40847322 Free PMC article.
-
Combination of mesenchymal stem cells and three-dimensional collagen scaffold preserves ventricular remodeling in rat myocardial infarction model.World J Stem Cells. 2022 Aug 26;14(8):633-657. doi: 10.4252/wjsc.v14.i8.633. World J Stem Cells. 2022. PMID: 36157910 Free PMC article.
References
-
- Lee T.Y., Chin G.S., Kim W.J., Chau D., Gittes G.K., and Longaker M.T.. Expression of transforming growth factor beta 1, 2, and 3 proteins in keloids. Ann Plast Surg, 1999. 43(2): 179–184. - PubMed
-
- Qian H., Yang H., Xu W., Yan Y., Chen Q., Zhu W., et al. Bone marrow mesenchymal stem cells ameliorate rat acute renal failure by differentiation into renal tubular epithelial-like cells. Int J Mol Med, 2008. 22(3): 325–332. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

