Determination of a Key Antigen for Immunological Intervention To Target the Latent Stage of Toxoplasma gondii
- PMID: 28446567
- PMCID: PMC5473426
- DOI: 10.4049/jimmunol.1700062
Determination of a Key Antigen for Immunological Intervention To Target the Latent Stage of Toxoplasma gondii
Abstract
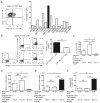
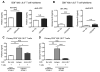
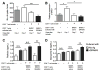
Toxoplasma gondii, an obligate intracellular protozoan parasite, establishes a chronic infection by forming cysts preferentially in the brain. Up to one third of the human population worldwide is estimated to be chronically infected with this parasite. However, there is currently no drug effective against the cyst form of the parasite. In addition, the protective immunity against the cysts remains largely unknown. We analyzed the molecular mechanisms by which the immune system detects host cells harboring the cysts to eliminate the latent stage of the parasite using mice with the H-2d haplotype, which are genetically resistant to the infection. Our study revealed that CD8+ immune T cells bearing TCR Vβ8.1, 8.2 chain have a potent activity to remove T. gondii cysts from the brain. Our studies also uncovered that H-2Ld is the major Ag-presenting molecule to CD8+ T cells for initiating cyst elimination, and that CD8+Vβ8.1, 8.2+ immune T cells recognize the N-terminal region (aa 41-152) of dense granule protein 6 (GRA6Nt) of the parasite presented by the H-2Ld molecule. Furthermore, CD8+ immune T cells induced by immunization with recombinant GRA6Nt were eventually capable of removing the cysts from the brain when transferred to infected immunodeficient mice lacking T cells. Thus, GRA6Nt is a novel and potent Ag to activate CD8+ T cells capable of removing T. gondii cysts. These observations offer a basis for immunological intervention to combat chronic infection with T. gondii by targeting the persistent cysts of the parasite.
Copyright © 2017 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures


References
-
- Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965–1976. - PubMed
-
- Munoz M, Liesenfeld O, Heimesaat MM. Immunology of Toxoplasma gondii. Immunol Rev. 2011;240:269–285. - PubMed
-
- Suzuki Y, Orellana MA, Schreiber RD, Remington JS. Interferon-gamma: the major mediator of resistance against Toxoplasma gondii. Science. 1988;240:516–518. - PubMed
-
- Israelski DM, Remington JS. Toxoplasmosis in the non-AIDS immunocompromised host. In: Remington JS, Swrltz M, editors. Curr Clin Top Infect Dis. Blackwell Scientific Publications; London: 1993. pp. 322–356. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

