Change in Diabetic Retinopathy Through 2 Years: Secondary Analysis of a Randomized Clinical Trial Comparing Aflibercept, Bevacizumab, and Ranibizumab
- PMID: 28448655
- PMCID: PMC5540030
- DOI: 10.1001/jamaophthalmol.2017.0821
Change in Diabetic Retinopathy Through 2 Years: Secondary Analysis of a Randomized Clinical Trial Comparing Aflibercept, Bevacizumab, and Ranibizumab
Abstract
Importance: Anti-vascular endothelial growth factor (anti-VEGF) therapy for diabetic macular edema (DME) favorably affects diabetic retinopathy (DR) improvement and worsening. It is unknown whether these effects differ across anti-VEGF agents.
Objective: To compare changes in DR severity during aflibercept, bevacizumab, or ranibizumab treatment for DME.
Design, setting, and participants: Preplanned secondary analysis of data from a comparative effectiveness trial for center-involved DME was conducted in 650 participants receiving aflibercept, bevacizumab, or ranibizumab. Retinopathy improvement and worsening were determined during 2 years of treatment. Participants were randomized in 2012 through 2013, and the trial concluded on September 23, 2015.
Interventions: Random assignment to aflibercept, 2.0 mg; bevacizumab, 1.25 mg; ranibizumab, 0.3 mg, up to every 4 weeks through 2 years following a retreatment protocol.
Main outcomes and measures: Percentages with retinopathy improvement at 1 and 2 years and cumulative probabilities for retinopathy worsening through 2-year without adjustment for multiple outcomes.
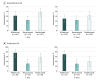
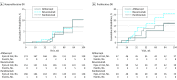
Results: A total of 650 participants (495 [76.2%] nonproliferative DR [NPDR], 155 proliferative DR [PDR]) were analyzed; 302 (46.5%) were women and mean (SD) age was 61 (10) years; 425 (65.4%) were white. At 1 year, among 423 NPDR eyes, 44 of 141 (31.2%) treated with aflibercept, 29 of 131 (22.1%) with bevacizumab, and 57 of 151 (37.7%) with ranibizumab had improvement of DR severity (adjusted difference: 11.7%; 95% CI, 2.9% to 20.6%; P = .004 for aflibercept vs bevacizumab; 8.9%; 95% CI, 1.7% to 16.1%; P = .01 for ranibizumab vs bevacizumab; and 2.9%; 95% CI, -5.7% to 11.4%; P = .51 for aflibercept vs ranibizumab). At 2 years, 33 eyes (24.8%) in the aflibercept group, 25 eyes (22.1%) in the bevacizumab group, and 40 eyes (31.0%) in the ranibizumab group had DR improvement; no treatment group differences were identified. For 93 eyes with PDR at baseline, 1-year improvement rates were 75.9% for aflibercept, 31.4% for bevacizumab, and 55.2% for ranibizumab (adjusted difference: 50.4%; 95% CI, 26.8% to 74.0%; P < .001 for aflibercept vs bevacizumab; 20.4%; 95% CI, -3.1% to 44.0%; P = .09 for ranibizumab vs bevacizumab; and 30.0%; 95% CI, 4.4% to 55.6%; P = .02 for aflibercept vs ranibizumab). These rates and treatment group differences appeared to be maintained at 2 years. Despite the reduced numbers of injections in the second year, 66 (59.5%) of NPDR and 28 (70.0%) of PDR eyes that manifested improvement at 1 year maintained improvement at 2 years. Two-year cumulative rates for retinopathy worsening ranged from 7.1% to 10.2% and 17.2% to 26.4% among anti-VEGF groups for NPDR and PDR eyes, respectively. No statistically significant treatment differences were noted.
Conclusions and relevance: At 1 and 2 years, eyes with NPDR receiving anti-VEGF treatment for DME may experience improvement in DR severity. Less improvement was demonstrated with bevacizumab at 1 year than with aflibercept or ranibizumab. Aflibercept was associated with more improvement at 1 and 2 years in the smaller subgroup of participants with PDR at baseline. All 3 anti-VEGF treatments were associated with low rates of DR worsening. These data provide additional outcomes that might be considered when choosing an anti-VEGF agent to treat DME.
Conflict of interest statement
Figures
Comment in
-
Diabetic Retinopathy and Antivascular Endothelial Growth Factor Agents.JAMA Ophthalmol. 2017 Jun 1;135(6):568-569. doi: 10.1001/jamaophthalmol.2017.0318. JAMA Ophthalmol. 2017. PMID: 28448646 No abstract available.
References
-
- Stone TW. ASRS 2015 Preferences and Trends Membership Survey. Chicago, IL: American Society of Retina Specialists; 2015.
-
- Korobelnik JF, Do DV, Schmidt-Erfurth U, et al. Intravitreal aflibercept for diabetic macular edema. Ophthalmology. 2014;121(11):2247-2254. - PubMed
-
- Googe J, Brucker AJ, Bressler NM, et al. ; Diabetic Retinopathy Clinical Research Network . Randomized trial evaluating short-term effects of intravitreal ranibizumab or triamcinolone acetonide on macular edema after focal/grid laser for diabetic macular edema in eyes also receiving panretinal photocoagulation. Retina. 2011;31(6):1009-1027. - PMC - PubMed
-
- Nguyen QD, Brown DM, Marcus DM, et al. ; RISE and RIDE Research Group . Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789-801. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

