Functions of galectins as 'self/non-self'-recognition and effector factors
- PMID: 28449072
- PMCID: PMC5808656
- DOI: 10.1093/femspd/ftx046
Functions of galectins as 'self/non-self'-recognition and effector factors
Abstract
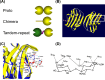
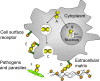
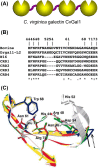
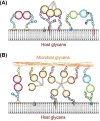
Carbohydrate structures on the cell surface encode complex information that through specific recognition by carbohydrate-binding proteins (lectins) modulates interactions between cells, cells and the extracellular matrix, or mediates recognition of potential microbial pathogens. Galectins are a family of ß-galactoside-binding lectins, which are evolutionary conserved and have been identified in most organisms, from fungi to invertebrates and vertebrates, including mammals. Since their discovery in the 1970s, their biological roles, initially understood as limited to recognition of endogenous carbohydrate ligands in embryogenesis and development, have expanded in recent years by the discovery of their roles in tissue repair and regulation of immune homeostasis. More recently, evidence has accumulated to support the notion that galectins can also bind glycans on the surface of potentially pathogenic microbes, and function as recognition and effector factors in innate immunity, thus establishing a new paradigm. Furthermore, some parasites 'subvert' the recognition roles of the vector/host galectins for successful attachment or invasion. These recent findings have revealed a striking functional diversification in this structurally conserved lectin family.
Keywords: carbohydrate recognition domain: glycans; chimera; function; galectins, ß-galactoside; pattern recognition receptors; proto-type; structure; tandem repeat.
© FEMS 2017. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
References
-
- Ahmed H, Banerjee PP, Vasta GR. Differential expression of galectins in normal, benign and malignant prostate epithelial cells: silencing of galectin-3 expression in prostate cancer by its promoter methylation. Biochem Bioph Res Co 2007;358:241–6. - PubMed
-
- Ahmed H, Bianchet MA, Amzel LM et al. Novel carbohydrate specificity of the 16-kDa galectin from Caenorhabditis elegans: binding to blood group precursor oligosaccharides (type 1, type 2, Talpha, and Tbeta) and gangliosides. Glycobiology 2002;12:451–61. - PubMed
-
- Ahmed H, Du S-J, O’Leary N et al. Biochemical and molecular characterization of galectins from zebrafish (Danio rerio): notochord-specific expression of a prototype galectin during early embryogenesis. Glycobiology 2004;14:219–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

