Small non-coding RNA and cancer
- PMID: 28449079
- PMCID: PMC6248440
- DOI: 10.1093/carcin/bgx026
Small non-coding RNA and cancer
Abstract
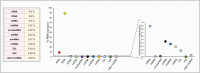
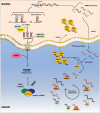
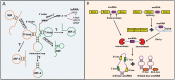
The ENCODE project has reported that at least 80% of the human genome is biologically active, yet only a small part of human DNA encodes for protein. The massive amount of RNA transcribed but not translated into protein can be classified as housekeeping RNA (such as rRNA, tRNA) and regulatory RNA (such as miRNA, piRNA, lncRNA). Small non-coding RNAs, in particular, have been the focus of many studies in the last 20 years and their fundamental role in many human diseases is currently well established. Inter alia, their role in cancer development and progression, as well as in drug resistance, is being increasingly investigated. In this review, focusing our attention on recent research results, we provide an overview of the four large classes of small non-coding RNAs, namely, miRNAs, piRNAs, snoRNA and the new class of tRNA-derived fragments, highlighting their fundamental role in cancer and their potential as diagnostic and prognostic biomarkers.
© The Author 2017. Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures
References
-
- Elgar G., et al. (2008) Tuning in to the signals: noncoding sequence conservation in vertebrate genomes. Trends Genet., 24, 344–352. - PubMed
-
- Pennisi E. (2012) Genomics. ENCODE project writes eulogy for junk DNA. Science, 337, 1159–1161. - PubMed
-
- Acunzo M., et al. (2015) MicroRNA and cancer–a brief overview. Adv. Biol. Regul., 57, 1–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

