The Abundance of Endofungal Bacterium Rhizobium radiobacter (syn. Agrobacterium tumefaciens) Increases in Its Fungal Host Piriformospora indica during the Tripartite Sebacinalean Symbiosis with Higher Plants
- PMID: 28450855
- PMCID: PMC5390018
- DOI: 10.3389/fmicb.2017.00629
The Abundance of Endofungal Bacterium Rhizobium radiobacter (syn. Agrobacterium tumefaciens) Increases in Its Fungal Host Piriformospora indica during the Tripartite Sebacinalean Symbiosis with Higher Plants
Abstract
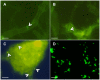
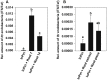
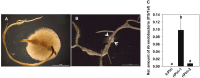
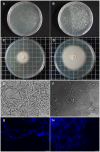
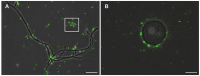
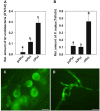
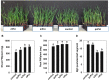
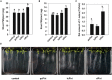
Rhizobium radiobacter (syn. Agrobacterium tumefaciens, syn. "Agrobacterium fabrum") is an endofungal bacterium of the fungal mutualist Piriformospora (syn. Serendipita) indica (Basidiomycota), which together form a tripartite Sebacinalean symbiosis with a broad range of plants. R. radiobacter strain F4 (RrF4), isolated from P. indica DSM 11827, induces growth promotion and systemic resistance in cereal crops, including barley and wheat, suggesting that R. radiobacter contributes to a successful symbiosis. Here, we studied the impact of endobacteria on the morphology and the beneficial activity of P. indica during interactions with plants. Low numbers of endobacteria were detected in the axenically grown P. indica (long term lab-cultured, lcPiri) whereas mycelia colonizing the plant root contained increased numbers of bacteria. Higher numbers of endobacteria were also found in axenic cultures of P. indica that was freshly re-isolated (riPiri) from plant roots, though numbers dropped during repeated axenic re-cultivation. Prolonged treatments of P. indica cultures with various antibiotics could not completely eliminate the bacterium, though the number of detectable endobacteria decreased significantly, resulting in partial-cured P. indica (pcPiri). pcPiri showed reduced growth in axenic cultures and poor sporulation. Consistent with this, pcPiri also showed reduced plant growth promotion and reduced systemic resistance against powdery mildew infection as compared with riPiri and lcPiri. These results are consistent with the assumption that the endobacterium R. radiobacter improves P. indica's fitness and thus contributes to the success of the tripartite Sebacinalean symbiosis.
Keywords: P. Indica; endobacteria; endofungal bacteria; endophytes; plant growth promotion bacteria; root colonization; tripartite symbiosis.
Figures
Similar articles
-
Non-pathogenic Rhizobium radiobacter F4 deploys plant beneficial activity independent of its host Piriformospora indica.ISME J. 2016 Apr;10(4):871-84. doi: 10.1038/ismej.2015.163. Epub 2015 Oct 23. ISME J. 2016. PMID: 26495996 Free PMC article.
-
NPR1 is required for root colonization and the establishment of a mutualistic symbiosis between the beneficial bacterium Rhizobium radiobacter and barley.Environ Microbiol. 2021 Apr;23(4):2102-2115. doi: 10.1111/1462-2920.15356. Epub 2020 Dec 22. Environ Microbiol. 2021. PMID: 33314556
-
The Piriformospora indica effector PIIN_08944 promotes the mutualistic Sebacinalean symbiosis.Front Plant Sci. 2015 Oct 26;6:906. doi: 10.3389/fpls.2015.00906. eCollection 2015. Front Plant Sci. 2015. PMID: 26579156 Free PMC article.
-
Piriformospora indica-a mutualistic basidiomycete with an exceptionally large plant host range.Mol Plant Pathol. 2012 Jun;13(5):508-18. doi: 10.1111/j.1364-3703.2011.00764.x. Epub 2011 Nov 24. Mol Plant Pathol. 2012. PMID: 22111580 Free PMC article. Review.
-
Endofungal Bacteria Increase Fitness of their Host Fungi and Impact their Association with Crop Plants.Curr Issues Mol Biol. 2019;30:59-74. doi: 10.21775/cimb.030.059. Epub 2018 Aug 2. Curr Issues Mol Biol. 2019. PMID: 30070651 Review.
Cited by
-
Serendipita indica E5'NT modulates extracellular nucleotide levels in the plant apoplast and affects fungal colonization.EMBO Rep. 2019 Feb;20(2):e47430. doi: 10.15252/embr.201847430. Epub 2019 Jan 14. EMBO Rep. 2019. PMID: 30642845 Free PMC article.
-
A Poly(A) Ribonuclease Controls the Cellotriose-Based Interaction between Piriformospora indica and Its Host Arabidopsis.Plant Physiol. 2018 Mar;176(3):2496-2514. doi: 10.1104/pp.17.01423. Epub 2018 Jan 25. Plant Physiol. 2018. PMID: 29371249 Free PMC article.
-
Meroterpenoids Possibly Produced by a Bacterial Endosymbiont of the Tropical Basidiomycete Echinochaete brachypora.Biomolecules. 2022 May 28;12(6):755. doi: 10.3390/biom12060755. Biomolecules. 2022. PMID: 35740880 Free PMC article.
-
Bacteria Community Inhabiting Heterobasidion Fruiting Body and Associated Wood of Different Decay Classes.Front Microbiol. 2022 May 3;13:864619. doi: 10.3389/fmicb.2022.864619. eCollection 2022. Front Microbiol. 2022. PMID: 35591994 Free PMC article.
-
Endofungal Rhizobium species enhance arsenic tolerance in colonized host plant under arsenic stress.Arch Microbiol. 2022 Jun 8;204(7):375. doi: 10.1007/s00203-022-02972-0. Arch Microbiol. 2022. PMID: 35674927
References
-
- Anca I. A., Lumini E., Ghignone S., Salvioli A., Bianciotto V., Bonfante P. (2009). The ftsZ gene of the endocellular bacterium ‘Candidatus Glomeribacter gigasporaum’ is preferentially expressed during the symbiotic phases of its host mycorrhizal fungus. Mol. Plant Microbe Interact. 22 302–310. 10.1094/MPMI-22-3-0302 - DOI - PubMed
-
- Bertaux J., Schmid M., Prevost-Bourre N. C., Churin J. L., Hartmann A., Garbaye J., et al. (2003). In situ identification of intracellular bacteria related to Paenibacillus spp. in the mycelium of the ectomycorrhizal fungus Laccaria bicolor S238N. Appl. Environ. Microbiol. 69 4243–4248. 10.1128/AEM.69.7.4243-4248.2003 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

