Exploring secondary-sphere interactions in Fe-N x H y complexes relevant to N2 fixation
- PMID: 28451336
- PMCID: PMC5363375
- DOI: 10.1039/c6sc04805f
Exploring secondary-sphere interactions in Fe-N x H y complexes relevant to N2 fixation
Abstract
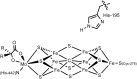
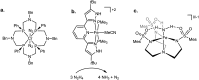
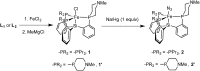
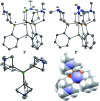
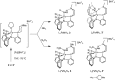
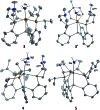
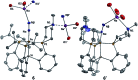
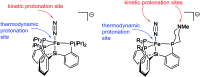
Hydrogen bonding and other types of secondary-sphere interactions are ubiquitous in metalloenzyme active sites and are critical to the transformations they mediate. Exploiting secondary sphere interactions in synthetic catalysts to study the role(s) they might play in biological systems, and to develop increasingly efficient catalysts, is an important challenge. Whereas model studies in this broad context are increasingly abundant, as yet there has been relatively little progress in the area of synthetic catalysts for nitrogen fixation that incorporate secondary sphere design elements. Herein we present our first study of Fe-N x H y complexes supported by new tris(phosphine)silyl ligands, abbreviated as [SiPNMe3] and [SiPiPr2PNMe], that incorporate remote tertiary amine hydrogen-bond acceptors within a tertiary phosphine/amine 6-membered ring. These remote amine sites facilitate hydrogen-bonding interactions via a boat conformation of the 6-membered ring when certain nitrogenous substrates (e.g., NH3 and N2H4) are coordinated to the apical site of a trigonal bipyramidal iron complex, and adopt a chair conformation when no H-bonding is possible (e.g., N2). Countercation binding at the cyclic amine is also observed for anionic {Fe-N2}- complexes. Reactivity studies in the presence of proton/electron sources show that the incorporated amine functionality leads to rapid generation of catalytically inactive Fe-H species, thereby substantiating a hydride termination pathway that we have previously proposed deactivates catalysts of the type [EPR3]FeN2 (E = Si, C).
Figures
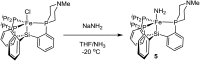
References
-
-
Selected reviews and examples:
- Holm R. H., Solomon E. I. Chem. Rev. 2004;104:347. - PubMed
- Cook S. A., Borovik A. S. Acc. Chem. Res. 2015;48:2407. - PMC - PubMed
- Cramer C. J., Tolman W. B. Acc. Chem. Res. 2007;40:601. - PMC - PubMed
- Costas M., Mehn M. P., Jensen M. P., Que, Jr. L. Chem. Rev. 2004;104:939. - PubMed
- Ohta S., Ohki Y., Hashimoto T., Cramer R. E., Tatsumi K. Inorg. Chem. 2012;51:11217. - PubMed
- Collman J. P., Boulatov R., Sunderland C. J., Fu L. Chem. Rev. 2004;104:561. - PubMed
- Tsui E. Y., Kanady J. S., Agapie T. Inorg. Chem. 2013;52:13833. - PMC - PubMed
- Chatt J., Leigh G. J. Chem. Soc. Rev. 1972;1:121.
- Kovacs J. A., Brines L. M. Acc. Chem. Res. 2007;40:501. - PMC - PubMed
- Itoh S. Acc. Chem. Res. 2015;48:2066. - PubMed
- Hematian S., Garcia-Bosch I., Karlin K. D. Acc. Chem. Res. 2015;48:2462. - PMC - PubMed
- Lyons C. T., Stack T. D. P. Coord. Chem. Rev. 2013;257:528. - PMC - PubMed
- Citek C., Herres-Pawlis S., Stack T. D. P. Acc. Chem. Res. 2015;48:2424. - PubMed
- Serrano-Plana J., Garcia-Bosch I., Company A., Costas M. Acc. Chem. Res. 2015;48:2397. - PubMed
- Friedle S., Reisner E., Lippard S. J. Chem. Soc. Rev. 2010;39:2768. - PMC - PubMed
-
-
-
Selected reviews and examples:
- Dutta A., Roberts J. A. S., Shaw W. J. Angew. Chem., Int. Ed. 2014;53:6487. - PubMed
- Henry R. M., Shoemaker R. K., DuBois D. L., Rakowski DuBois M. J. Am. Chem. Soc. 2006;128:3002. - PubMed
- Rakowski DuBois M., DuBois D. L. Chem. Soc. Rev. 2009;38:62. - PubMed
- Rauchfuss T. B. Acc. Chem. Res. 2015;48:2107. - PMC - PubMed
- Shook R. L., Borovik A. S. Inorg. Chem. 2010;49:3646. - PMC - PubMed
- MacBeth C. E., Golombek A. P., Young V. G., Yang C., Kuczera K., Hendrich M. P., Borovik A. S. Science. 2000;289:938. - PubMed
- MacBeth C. E., Gupta R., Mitchell-Koch K. R., Young V. G., Lushington G. H., Thompson W. H., Hendrich M. P., Borovik A. S. J. Am. Chem. Soc. 2004;126:2556. - PubMed
- Lacy D. C., Gupta R., Stone K. L., Greaves J., Ziller J. W., Hendrich M. P., Borovik A. S. J. Am. Chem. Soc. 2010;132:12188. - PMC - PubMed
- Carver C. T., Matson B. D., Mayer J. M. J. Am. Chem. Soc. 2012;134:5444. - PubMed
- Constentin C., Drouet S., Robert M., Saveant J.-M. Science. 2012;338:90. - PubMed
- Chang C. J., Chng L. L., Nocera D. G. J. Am. Chem. Soc. 2003;125:1866. - PubMed
- Lee C. H., Dogutan D. K., Nocera D. G. J. Am. Chem. Soc. 2011;133:8775. - PubMed
- Soo H. S., Komor A. C., Iavarone A. T., Chang C. J. Inorg. Chem. 2009;48:10024. - PubMed
- Matson E. M., Bertke J. A., Fout A. R. Inorg. Chem. 2014;53:4450. - PubMed
- Matson E. M., Park Y. J., Fout A. R. J. Am. Chem. Soc. 2014;136:17398. - PubMed
- Wallen C. M., Palatinus L., Bacsa J., Scarborough C. C. Angew. Chem., Int. Ed. 2016;55:11902. - PubMed
-
-
-
Selected reviews and examples:
- Holm R. H., Kennepohl P., Solomon E. I. Chem. Rev. 1996;96:2239. - PubMed
- Lu Y., Valentine J. S. Curr. Opin. Struct. Biol. 1997;7:495. - PubMed
- Kiefer L. L., Paterno S. A., Fierke C. A. J. Am. Chem. Soc. 1995;117:6831.
- Vogt L., Vinyard D. J., Khan S., Brudvig G. W. Curr. Opin. Chem. Biol. 2015;25:152. - PubMed
- Kung Y., Drennan C. L. Curr. Opin. Chem. Biol. 2011;15:276. - PMC - PubMed
- Moore C. M., Dahl E. W., Szymczak N. K. Curr. Opin. Chem. Biol. 2015;25:9. - PubMed
- Fontecilla-Camps J. C., Volbeda A., Cavazza C., Nicolet Y. Chem. Rev. 2007;107:4273. - PubMed
- Fernandez D., Pallares I., Vendrell J., Aviles F. X. Biochimie. 2010;92:1484. - PubMed
- Galinato M. G. I., Spolitak T., Ballou D. P., Lehnert N. Biochemistry. 2011;50:1053. - PMC - PubMed
- Cook S. A., Hill E. A., Borovik A. S. Biochemistry. 2015;54:4167. - PMC - PubMed
-
-
- Hoffman B. M., Dean D. R., Seefeldt L. C. Acc. Chem. Res. 2009;42:609. - PMC - PubMed
- Hoffman B. M., Lukoyanov D., Dean D. R., Seefeldt L. C. Acc. Chem. Res. 2013;46:587. - PMC - PubMed
- Dos Santos P. C., Igarashi R. Y., Lee H.-I., Hoffman B. M., Seefeldt L. C., Dean D. R. Acc. Chem. Res. 2005;38:208. - PubMed
-
- Barney B. M., Igarashi R. Y., Dos Santos P. C., Dean D. R., Seefeldt L. C. J. Biol. Chem. 2004;279:53621. - PubMed
- Seefeldt L. C., Dance I. G., Dean D. R. Biochemistry. 2004;43:1401. - PubMed
- Siegbahn P. E. M. J. Am. Chem. Soc. 2016;138:10485. - PubMed
- Schimpl J., Petrilli H. M., Blochl P. E. J. Am. Chem. Soc. 2003;125:15772. - PubMed
- Hallmen P. P., Kastner J. Z. Anorg. Allg. Chem. 2015;641:118.
- Kastner J., Hemmen S., Blochl P. E. J. Chem. Phys. 2005;123:074306. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

