Increased gait variability may not imply impaired stride-to-stride control of walking in healthy older adults: Winner: 2013 Gait and Clinical Movement Analysis Society Best Paper Award
- PMID: 28454071
- PMCID: PMC5869351
- DOI: 10.1016/j.gaitpost.2017.03.018
Increased gait variability may not imply impaired stride-to-stride control of walking in healthy older adults: Winner: 2013 Gait and Clinical Movement Analysis Society Best Paper Award
Abstract
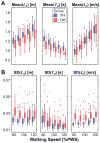
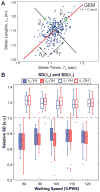
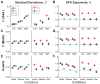
Older adults exhibit increased gait variability that is associated with fall history and predicts future falls. It is not known to what extent this increased variability results from increased physiological noise versus a decreased ability to regulate walking movements. To "walk", a person must move a finite distance in finite time, making stride length (Ln) and time (Tn) the fundamental stride variables to define forward walking. Multiple age-related physiological changes increase neuromotor noise, increasing gait variability. If older adults also alter how they regulate their stride variables, this could further exacerbate that variability. We previously developed a Goal Equivalent Manifold (GEM) computational framework specifically to separate these causes of variability. Here, we apply this framework to identify how both young and high-functioning healthy older adults regulate stepping from each stride to the next. Healthy older adults exhibited increased gait variability, independent of walking speed. However, despite this, these healthy older adults also concurrently exhibited no differences (all p>0.50) from young adults either in how their stride variability was distributed relative to the GEM or in how they regulated, from stride to stride, either their basic stepping variables or deviations relative to the GEM. Using a validated computational model, we found these experimental findings were consistent with increased gait variability arising solely from increased neuromotor noise, and not from changes in stride-to-stride control. Thus, age-related increased gait variability likely precedes impaired stepping control. This suggests these changes may in turn precede increased fall risk.
Keywords: Motor control; Older adults; Stepping; Variability; Walking.
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures

References
-
- Kannus P, Sievänen H, Palvanen M, Järvinen T, Parkkari J. Prevention of falls and consequent injuries in elderly people. Lancet. 2005;366:1885–93. - PubMed
-
- Sterling DA, O’Connor JA, Bonadies J. Geriatric falls: Injury severity is high and disproportionate to mechanism. Journal of Trauma. 2001;50:116–9. - PubMed
-
- Stevens JA, Mack KA, Paulozzi LJ, Ballesteros MF. Self-Reported Falls and Fall-Related Injuries Among Persons Aged ≥ 65 Years–United States, 2006. Journal of Safety Research. 2008;39:345–9. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

