Multi-site Neurogenin3 Phosphorylation Controls Pancreatic Endocrine Differentiation
- PMID: 28457793
- PMCID: PMC5425251
- DOI: 10.1016/j.devcel.2017.04.004
Multi-site Neurogenin3 Phosphorylation Controls Pancreatic Endocrine Differentiation
Abstract
The proneural transcription factor Neurogenin3 (Ngn3) plays a critical role in pancreatic endocrine cell differentiation, although regulation of Ngn3 protein is largely unexplored. Here we demonstrate that Ngn3 protein undergoes cyclin-dependent kinase (Cdk)-mediated phosphorylation on multiple serine-proline sites. Replacing wild-type protein with a phosphomutant form of Ngn3 increases α cell generation, the earliest endocrine cell type to be formed in the developing pancreas. Moreover, un(der)phosphorylated Ngn3 maintains insulin expression in adult β cells in the presence of elevated c-Myc and enhances endocrine specification during ductal reprogramming. Mechanistically, preventing multi-site phosphorylation enhances both Ngn3 stability and DNA binding, promoting the increased expression of target genes that drive differentiation. Therefore, multi-site phosphorylation of Ngn3 controls its ability to promote pancreatic endocrine differentiation and to maintain β cell function in the presence of pro-proliferation cues and could be manipulated to promote and maintain endocrine differentiation in vitro and in vivo.
Keywords: diabetes; endocrine differentiation; insulinoma; neurogenin3; pancreatic development; pancreatic organoids; proneural bHLH transcription factors; β cells.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures





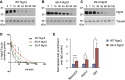
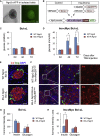
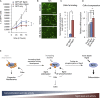
Comment in
-
β Cell Renewal versus Differentiation: Slow and Steady Wins the Race.Dev Cell. 2017 May 8;41(3):223-225. doi: 10.1016/j.devcel.2017.04.017. Dev Cell. 2017. PMID: 28486127
References
-
- Agudo J., Ayuso E., Jimenez V., Salavert A., Casellas A., Tafuro S., Haurigot V., Ruberte J., Segovia J.C., Bueren J. IGF-I mediates regeneration of endocrine pancreas by increasing beta cell replication through cell cycle protein modulation in mice. Diabetologia. 2008;51:1862–1872. - PubMed
-
- Al-Hasani K., Pfeifer A., Courtney M., Ben-Othman N., Gjernes E., Vieira A., Druelle N., Avolio F., Ravassard P., Leuckx G. Adult duct-lining cells can reprogram into beta-like cells able to counter repeated cycles of toxin-induced diabetes. Dev. Cell. 2013;26:86–100. - PubMed
-
- Ali F.R., Cheng K., Kirwan P., Metcalfe S., Livesey F.J., Barker R.A., Philpott A. The phosphorylation status of Ascl1 is a key determinant of neuronal differentiation and maturation in vivo and in vitro. Development. 2014;141:2216–2224. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 098357/Z/12/Z /WT_/Wellcome Trust/United Kingdom
- 22585/CRUK_/Cancer Research UK/United Kingdom
- 19013/CRUK_/Cancer Research UK/United Kingdom
- 1509287/MRC_/Medical Research Council/United Kingdom
- MR/K018329/1/MRC_/Medical Research Council/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- MR/M008975/1/MRC_/Medical Research Council/United Kingdom
- MC_PC_12009/MRC_/Medical Research Council/United Kingdom
- 12077/CRUK_/Cancer Research UK/United Kingdom
- 097922/Z/11/Z/WT_/Wellcome Trust/United Kingdom
- MR/L021129/1/MRC_/Medical Research Council/United Kingdom
- 104151/Z/14/A /WT_/Wellcome Trust/United Kingdom
- 17044/CRUK_/Cancer Research UK/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

